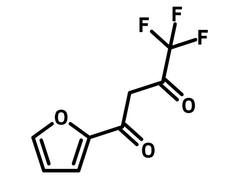
2-Furoyltrifluoroacetone
CAS Number 326-90-9
Fluorinated Building Blocks, Heterocyclic Building Blocks, Monomers, Chemistry Building Blocks,A fluorinated β-diketone building block
Used as a synthesis intermediate for metal complexes, isoxazoles, pyrimidine and pyrazoles in application of OLEDs, displays, and drug discovery
Specifications | MSDS | Literature and Reviews
2-Furoyltrifluoroacetone (CAS number 326-90-9) is a derivative of β-diketone with a furan and a trifluoromethyl substituents. 2-Furoyltrifluoroacetone is directly applied as a ligand to the synthesis of luminescent terbium (Tb III) and lanthanide (Ln III) complexes that are used OLEDs and displays. The complexes have high quantum efficiency of 77% and long lifetime of 842 μs.
The furan of 2-Furoyltrifluoroacetone can be easily modified via Diels-Alder reactions (with electron deficient dienophiles) and electrophilic substitution.
The β-diketone (1,3-diketone) moiety is a precursor for many other heterocyclic compounds. 2-Furoyltrifluoroacetone reacts with hydrazine to form pyrazole, with hydroxylamine to form isoxazole, and with urea pyrimidine. The heterocyclic compounds are investigated as active pharmaceutical ingredients (APIs) for anticancer and anti-inflammatory drug discovery.
Multiple functional groups
For facile synthesis
Fluorinated diketone building block
For drug discovery, ligands, and OLEDs
Worldwide shipping
Quick and reliable shipping
High purity
>98% High purity
General Information
| CAS Number | 326-90-9 |
| Chemical Formula | C8H5F3O3 |
| Full Name | 4,4,4-Trifluoro-1-(2-furyl)-1,3-butanedione |
| Molecular Weight | 206.12 g/mol |
| Synonyms | Perfluoroacetyl(2-furoyl)methane |
| Classification / Family | Fluorinated building block, Heterocyclic building block, Diketone Derivatives, Pyrazoles, OLEDs, APIs, Display |
Chemical Structure
Product Details
| Purity | 98% |
| Melting Point | Tm = 19 °C – 21 °C |
| Boiling Point | Tb= 203 °C at 760 mmHg |
| Relative Density | 1.391 g/mL at 25 °C |
| Appearance | Clear liquid |
MSDS Documentation
2-Furoyltrifluoroacetone MSDS Sheet
Literature and Reviews
- Luminescent Tb(III) complexes with Lewis bases for displays: Synthesis and spectral investigation, A. Hooda et al, Inorg. Chem. Commun., 151, 110583(2023); DOI: 10.1016/j.inoche.2023.110583.
- Anticancer activity of novel 3‐(furan‐2‐yl)pyrazolyl and 3‐(thiophen‐2‐yl)pyrazolyl hybrid chalcones: Synthesis and in vitro studies, M. Helmy et al., Arch. Pharm., 355, e2100381(2022); DOI: 10.1002/ardp.202100381.
- Exploration of photophysical behavior of lanthanide complex and its hybrids, S. Wankar et al., Spectrochim. Acta A Mol. Biomol., 254, 119629(2021); DOI: 10.1016/j.saa.2021.119629.
