Ethylammonium Bromide, EABr
CAS Number 593-55-5
Materials, Perovskite Materials, Perovskite Precursor MaterialsEthylammonium bromide, high purity perovskite precursor
Used to alter properties of perovskite materials to improve efficiency of hybrid electronic devices
Overview | Specifications | MSDS | Literature and Reviews
Ethylammonium bromide (EABr) has been used as a precursor for the alteration of crystallite structure, evolution of film morphology, and fine-tuning optical properties of perovskite materials to improve the efficiency of hybrid electronic devices.
Sky-blue layered perovskite light-emitting devices showing the maximum EQE of 2.6% and power efficiency of 1 lm/W at 100 cd/m2 have been achieved [1], with a device structure of ITO/PEDOT:PSS/ (EA)2(MA)n−1PbnBr3n+1 (50 nm)/TmPyPB (50 nm)/CsF/Al.
General Information
| CAS number | 593-55-5 |
| Chemical formula | C2H8BrN |
| Molecular weight | 125.99 g/mol |
| Synonyms | Ethylamine hydrobromide, Ethanamine hydrobromide, EABr |
| Classification / Family | Alkylammonium halides, Perovskite precursor materials, Perovskite solar cells, Perovskite LEDs |
Product Details
| Purity | >99% |
| Melting point | 160 °C |
| Color | Powder/crystals |
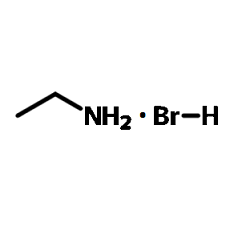
Chemical Structure
MSDS Documentation
Ethylammonium bromide MSDS sheet
Literature and Reviews
- Efficient Sky-Blue Perovskite Light-Emitting Devices Based on Ethylammonium Bromide Induced Layered Perovskites, Q. Wang et al., ACS Appl. Mater. Interfaces, 9 (35), 29901–29906 (2017); DOI: 10.1021/acsami.7b07458.
- Size of the Organic Cation Tunes the Band Gap of Colloidal Organolead Bromide Perovskite Nanocrystals, M, Mittal et al., J. Phys. Chem. Lett., 7 (16), 3270–3277 (2016); DOI: 10.1021/acs.jpclett.6b01406.
- Maximizing the emissive properties of Ch2Nh2PbBr3 perovskite nanoparticles, S. Gonzalez-Carrero et al., J. Mater. Chem. A, 3, 9187-9193 (2015); DOI: 10.1039/C4TA05878J.
