HDOQx-DBrDF
CAS Number 2269476-12-0
Chemistry Building Blocks, Dibromo Monomers, Fluorinated Building Blocks, Heterocyclic Building Blocks,HDOQx-DBrDF, high purity (>99%) monomer for PTQ10
Used in low band gap semiconducting polymers for organic photovoltaic devices
Specifications | MSDS | Literature and Reviews
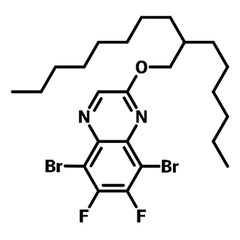
HDOQx-DBrDF, namely 5,8-Dibromo-6,7-difluoro-2-((2-hexyldecyl)oxy)quinoxaline (CAS number 2269476-12-0), is a fluorinated quinoxaline derivative. Quinoxaline derivatives are widely used as electron deficient units in low bandgap semiconducting polymers, such as PTQ10, for organic photovoltaic devices. Further introduction of fluorine atoms on the benzene ring makes electron density in the structure even poorer, downshifting the highest occupied molecular orbital (HOMO) energy level and increasing charge mobility of the targeted polymer donors. Fluorination also achieves fast charge separation and low nonradiative recombination loss in the PSCs, resulting both high VOC and JSC. Large hexyldecyloxy (HD) group is there to enhance absorption of the polymers and to improve the targeted polymers' solubility in most of the common solvents.
HDOQx-DBrDF has been used for the synthesis of PTQ10 as a highly efficient polymer semiconductor for NF-PSCs.
Capped with bromide
for facil coupling reactions
Worldwide shipping
Quick and reliable shipping
Quinoxaline building block
For semiconductors, OFETs, and solar cells
High purity
>99% High purity
General Information
| CAS Number | 2269476-12-0 |
| Chemical Formula | C24H34Br2F2N2O |
| Molecular Weight | 564.34 g/mol |
| Synonyms | 5,8-Dibromo-6,7-difluoro-2-((2-hexyldecyl)oxy)quinoxaline |
| Classification / Family | Quinoxaline, Semiconductor synthesis intermediates, Low band gap polymers, OFETs, Organic photovoltaics, polymer solar cells |
Chemical Structure
Product Details
| Purity | >99% (byHPLC, 254 nm, 1H NMR in CDCl3) |
| Melting Point | N/A |
| Appearance | Colorless oil |
MSDS Documentation
Literature and Reviews
- Achieving Fast Charge Separation and Low Nonradiative Recombination Loss by Rational Fluorination for High-Efficiency Polymer Solar Cells, C. Sun et al., Adv. Mater., 31, 1905480 (2019); DOI: 10.1002/adma.201905480.
- Exciton and Charge Carrier Dynamics in Highly Crystalline PTQ10:IDIC Organic Solar Cells, H. Cha et al., Adv. Energy Mater., 10 (38), 2001149 (2020); DOI: 10.1002/aenm.202001149.
- Tailored phase conversion under conjugated polymer enables thermally stable perovskite solar cells with efficiency exceeding 21%, L. Meng et al., J. Am. Chem. Soc., 140, 49, 17255–17262 (2018); DOI: 10.1021/jacs.8b10520.
