Wide Bandgap Polymer Semiconductor
Low cost polymer for doctor blading or inkjet printing device fabrication
Specifications | MSDS | Literature and Reviews | Technical Support
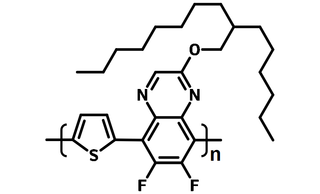
PTQ10 is a donor–acceptor (D–A) type copolymer with a simple thiophene ring as the electron donor unit and quinoxaline (QX) as electron acceptor unit. The alkoxy side chain attached on quinoxaline unit is to promote solubility and to enhance absorption of the targeted polymer. Difluoro substituted quinoxaline can further down-shift the HOMO energy level, achieve fast charge separation and low nonradiative recombination loss in the PSCs leading to high VOC and JSC.
PTQ10 can be processed in solution with eco-friendly non-halogenated solvents, i.e. o-xylene. The combined effects of high efficiency, low cost, and green solvents processibility makes PTQ10 an ideal candidate for large scale doctor-blading and inkjet printing device fabrications. Organic polymer solar cells based on PTQ10:Y6 blend shows efficient charge separation, higher photoluminescence quenching efficiency and higher charge mobilities, giving a power conversion efficiency of 16.53%.
Donor-Acceptor Copolymer
For high efficiency OPV devices
Large Batch Luminosyn™
High purity with batch-specific GPC data
Simple Worldwide Shipping
Reliable delivery via tracked courier
Green Energy Material
Process in non-halogenated solvents
The Luminosyn™ Range
General Information
| Full Name | Poly [[6,7-difluoro[(2-hexyldecyl)oxy]-5,8-quinoxalinediyl]-2,5-thiophenediyl ]] |
|---|---|
| Synonyms | PTQ10 |
| Chemical Formula | (C28H36F2N2OS)n |
| CAS Number | 2270233-86-6 |
| UV-Vis Absorption | λmax 556 nm, 600 nm in CB as-cast film |
| HOMO / LUMO | HOMO = -5.54 eV, LUMO = -2.98 eV [1] |
| Soluble In | o-xylene, chloroform, chlorobenzene and dichlorobenzene |
| Recommended Processing Solvents at 10mg/ml | o-xylene (8mg/ml), chloroform |
| Classification or Family | Organic semiconducting materials, Wide band-gap polymers, Organic Photovoltaics, Polymer solar cells, NF-PSCs, All-polymer solar cells (all-pscs), Clean energy materials. |
Batch Information
| Batch | Mw | Mn | PDI | Stock info |
|---|---|---|---|---|
| M2283A2 | 74.092 | 38,464 | 1.93 | In stock |
Chemical Structure
Device Structure
Device structure: ITO (indium tin oxide)/PEDOT:PSS AI 4083/PTQ10:Y6/PDINO/A1 [1]
| Thickness (nm) | VOC (V) | JSC (mA cm-2) | FF (%) | PCE (%) |
|---|---|---|---|---|
| 110 nm | 0.826 | 26.65 | 75.1 | 16.53 |
MSDS Documentation
Literature and Reviews
- Rationally pairing photoactive materials for high-performance polymer solar cells with efficiency of 16.53%, Y. Wu et al., Sci China Chem., 63: 265–271 (2020); DOI: 10.1007/s11426-019-9599-1.
- Achieving Fast Charge Separation and Low Nonradiative Recombination Loss by Rational Fluorination for High-Efficiency Polymer Solar Cells, C. Sun et al., Adv. Mater., 31, 1905480 (2019); DOI: 10.1002/adma.201905480.
- Exciton and Charge Carrier Dynamics in Highly Crystalline PTQ10:IDIC Organic Solar Cells, H. Cha et al., Adv. Energy Mater., 10 (38) 2001149 (2020); DOI: 10.1002/aenm.202001149.
