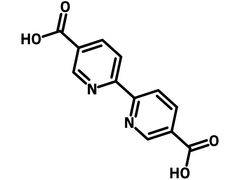
2,2′-Bipyridine-5,5′-dicarboxylic acid
CAS Number 1802-30-8
Chemistry Building Blocks, Heterocyclic Building Blocks, Materials, MOF Ligands,A frequent bipyridyl ligand with dicarboxylic acid anchoring groups
As the building block for the synthesis of organometallic compounds and the dicarboxylic acid anchoring groups are key functional groups to form 3D metal-organic frameworks (MOF)
Specifications | MSDS | Literature and Reviews
2,2'-Bipyridine-5,5'-dicarboxylic acid (CAS number 1802-30-8), an isomer to 2,2'-bipyridine-4,4'-dicarboxylic acid, is another bipyridyl structure with dicarboxylic acid anchoring groups at 5,-5'-postions. It is one of the most frequently used heterocyclic building blocks in the synthesis of organometallic compounds and the dicarboxylic acid anchoring groups are key functional groups to form 3D metal-organic frameworks (MOF) which find application in carbon dioxide reduction, organic photocatalysis, water oxidation, antioxidants and lithium- and sodium-ion batteries.
Due to the highly ordered stacking structure of 2,2′-bipyridine-5,5′-dicarboxylic acid (H2bpy) imposed by hydrogen bonding, 2,2′-bipyridine-5,5′-dicarboxylic acid can be employed as anode material showing high specific capacity, good cycle stability, and remarkable rate performance in both lithium and sodium-ion batteries. 2,2′-bipyridine-5,5′-dicarboxylic acid has been reported to possess an ultrahigh initial capacity of lithium-ion battery of 1200 mAh•g-1 at a current density of 200 mA•g-1, and the specific capacity can maintain 550 mAh•g-1 after 100 cycles.
Using 2,2'-bipyridine-5,5'-dicarboxylic acid as the binding ligand, both RuH(H2bpy)(PPh3)2(CO)] (I) and [RuH(H2bpy)(AsPh3)2(CO)] (II) shows excellent radical scavenging properties as potential cancer treating agents with less toxicity on normal cell NIH 3T3 and considerable cyto-toxic activity against HeLa and HepG2 cancer cell lines.
Bipyridine building block
as a ligand for porous organic frameworks
Worldwide shipping
Quick and reliable shipping
Multiple functionalities
used in batteries, photocatalysis and medicinal chemistry
High purity
>98% Purity
General Information
| CAS Number | 1802-30-8 |
| Chemical Formula | C12H8N2O4 |
| Full Name | 2,2′-Bipyridine-5,5′-dicarboxylic acid |
| Molecular Weight | 244.20 g/mol |
| Synonyms | 5,5'-Dcbpy, 2,2′-Bipyridyl-5,5′-dicarboxylic acid, 6,6′-Binicotinic acid |
| Classification / Family | Bipyridyl derivatives, Semiconductor synthesis intermediates, Dye-sensitized solar cells (DSSCs) |
Chemical Structure
Product Details
| Purity | >98% (1H NMR) |
| Melting Point | >365 °C |
| Appearance | White off-white to orange powder/crystals |
MSDS Documentation
2,2′-Bipyridine-5,5′-dicarboxylic acid MSDS Sheet
Literature and Reviews
-
Bipyridine carboxylic acid as a high-performance anode material for lithium- and sodium-ion batteries, Y. Bo et al., Electrochim. Acta, 405, 139628 (2022); DOI: 10.1016/j.electacta.2021.139628.
-
Cationic Zr-based metal-organic framework via post-synthetic alkylation for selective adsorption and separation of anionic dyes, J. Liu et al., Mater. Today Chem., 24, 100897 (2022); DOI: 10.1016/j.mtchem.2022.100897.
-
Protection Against Cu(II)-Induced Oxidative Stress and Toxicity to Chlorella vulgaris by 2,2′-Bipyridine-5,5′-dicarboxylic Acid, Y. Wen et al., Arch. Environ. Contam. Toxicol. 66, 400–406 (2014); DOI: 10.1007/s00244-013-9977-2
