TCNQ
TCNQ, a powerful electron-accepting molecule
Helps increase hole mobility and the lowering of injection barriers
7,7,8,8-tetracyanoquinodimethane (TCNQ), CAS number 1518-16-7, with a LUMO at 4.5 eV, is known for the charge-transfer salts formed by its radical anion TCNQ in photovoltaic, light-emitting diodes, and organic field-effect transistor devices. TCNQ and its derivatives have been used as dopants, leading to an increase in hole mobility or to the lowering of injection barriers. One classic example of such is the treatment of tetrathiafulvene (TTF), an electron donor with TCNQ. TFF and TCNQ form an ion pair, the TTF-TCNQ complex. This process of doping leads to the crystallization of the ion pair into a one-dimensionally stacked polymer. This polymer consists of segregated stacks of cations and anions of the donors and the acceptors, respectively. The complex crystal is an organic semiconductor that exhibits metallic electric conductivity [1, 2].
It also has been shown that TCNQ can effectively modify Cu or Ag surfaces. The formation of Cu-TCNQ and Ag-TCNQ enhances the work function of such electrodes and reduces the hole injection barrier dramatically. Furthermore, it improves electrode/organic layer contact, hence the reduction of contact resistances [3].
Tetracyanoquinodimethane (TCNQ) is also found to act as a p-type doping agent of graphene films due to its powerful electron-accepting capacity [4].
General Information
| CAS number | 1518-16-7 |
|---|---|
| Chemical formula | C12H4N4 |
| Molecular weight | 204.19 g/mol |
| HOMO/LUMO | LUMO = 4.5 eV |
| Synonyms |
|
| Classification / Family | Electron acceptor, Hole-injection materials, Hole transport layer material, Light-emitting diodes, Polymer solar cells, OFETs. |
Product Details
| Purity | >99.0% (sublimed) |
| Melting point | 289 °C (dec.) |
|---|---|
| Appearance | Dark yellow to brown crystals/powder |
*Sublimation is a technique used to obtain ultra pure-grade chemicals. For more details about sublimation, please refer to the Sublimed Materials.
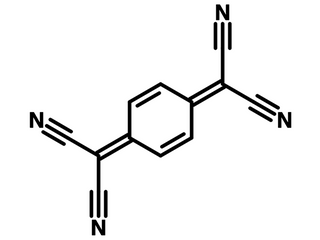
Chemical Structure
Device Structure(s)
| Device structure | ITO/0.4 wt% F4TCNQ doped α NPD (30 nm)/ 5 wt% Ir (ppy)3 doped CBP (50 nm)/BPhen (30 nm)/20 wt% TCNQ mixed BPhen (1.5 nm)/Al [5] |
|---|---|
| Color | Green |
| Luminance@15 V | 1,320 cd/m2 |
| Power Efficiency@14 V | 56.6 lm W−1 |
| Current Efficiency@14 V | 23.17 cd/A |
| Device structure | ITO/F4TCNQ (3 nm)/MeO-Spiro-TPD (27 nm)/CBP + BCzVbi* (50 nm)/BPhen (10 nm)/TCNQ mixed BPhen (1.5 nm)/Al [6] |
|---|---|
| Color | Red |
| Luminance@ 10 mA/cm2 | 1,790 cd/m2 |
| Power Efficiency@ 10 mA/cm2 | 4.65 lm W-1 |
| Current Efficiency@ 10 mA/cm2 | 18.0 cd/A |
*For chemical structure information, please refer to the cited references.
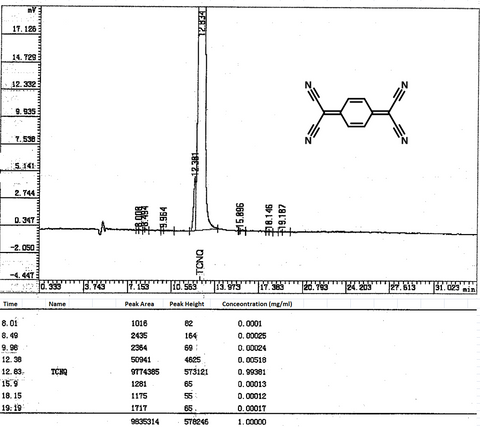
Characterization
Pricing
| Grade | Order Code | Quantity | Price |
|---|---|---|---|
| Sublimed (>99.0% purity) | M681 | 1 g | £165 |
| Sublimed (>99.0% purity) | M681 | 5 g | £680 |
MSDS Documentation
Literature and Reviews
- The Difference between Metallic and Insulating Salts of Tetracyanoquinodimethane (TCNQ): How to Design an Organic Metal, J. B. Torrance, Acc. Chem. Res., 12 (3), 79-86 (1979).
- Organic electronics: When TTF met TCNQ, J. R. Kirtley et al., Nat. Mater., 7, 520-521 (2008). doi:10.1038/nmat2211.
-
Interface Engineering: An Effective Approach toward High-Performance Organic Field-Effect
Transistors, C-A. Di et al., Acc. Chem. Res., 42 (10), 1573-1581 (2009).