D18-2Br
CAS Number 2433725-51-8
Dibromo Monomers, Heterocyclic Building Blocks, Monomers, Chemistry Building Blocks,Two electron-donating 5-bromo-4-butyloctylthiophene units
Used in the synthesis of polymer D18.
Specifications | MSDS | Literature and Reviews
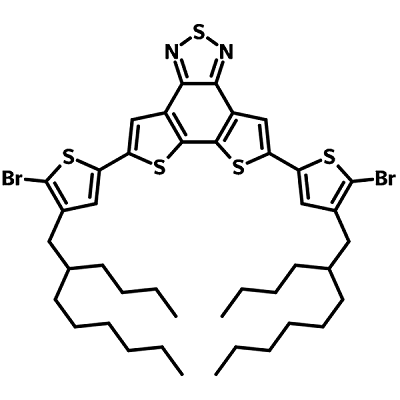
D18-2Br (CAS number 2433725-51-8) has a rigid electron accepting dithienobezothiadiazole (DTBT) core with two electron-donating 5-bromo-4-butyloctylthiophene units. The large side alkyl chains are there to promote solubility for solution processing of the end products that acquire D18-2Br as building block.
D18-2Br has been used for the synthesis of polymer D18 which gave a PCE of 18.22% while Y6 was used as the non-fullerene acceptor.
General Information
| CAS Number | 2433725-51-8 |
| Chemical Formula | C42H54Br2N2S5 |
| Molecular Weight | 907.02 g/mol |
| Full Name | 5,8-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3',2':3,4;2'',3'':5,6]benzo[1,2-c][1,2,5]thiadiazole |
| Synonyms | DTBT-2Th48Br |
| Classification / Family | Dithieno[3',2':3,4;2'',3'':5,6]benzo[1,2-c][1,2,5]thiadiazole (DTBT) derivatives, Organic semiconducting materials, Semiconductor Synthesis, Low band gap polymers, OFETs, OLED, Organic Photovoltaics, NFA-OSCs. |
Chemical Structure
Product Details
| Purity | >98% |
| Melting Point | N/A |
| Appearance | Orange powder/crystal |
MSDS Documentation
Literature and Reviews
- Multi-channel exciton dissociation in D18/Y6 complexes for high-efficiency organic photovoltaics, Z. Cao et al., J. Mater. Chem. A, 8, 20408-20413 (2020); doi: 10.1039/D0TA07996K.
- 18% Efficiency organic solar cells, Q Liu et al., Sci. Bull. 65, 272 (2020); doi: 10.1016/j.scib.2020.01.001.
- Nonfullerene Acceptors: A Renaissance in Organic Photovoltaics? P. Meredith et al., Adv. Energy Mater., 10 (33); 2001788 (2020); doi: 10.1002/aenm.202001788.
