IEICO-4Cl, low bandgap NFA with absorption into the near-infrared
Available for priority dispatch with free shipping on qualifying orders
Specifications | Pricing and Options | MSDS | Literature and Reviews
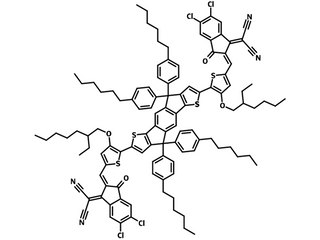
IEICO-4Cl (CAS number 2240998-88-1), a chlorinated version of IEICO-4F, has a structure with an electron-donating fused indaceno[1,2-b:5,6-b′]dithiophene (IDT) core flanked by two electron-withdrawing terminals, 2-(5,6-dichloro-3-oxo-2,3-dihydro-1H-inden-1-ylidene)malononitrile (IC-2Cl). IEICO-4Cl has an ultralow bandgap of 1.23 eV with weak absorption in the visible region, but extends its main absorption into the near-infrared (NIR) region (745-945 nm) with maximum absorption around 880 nm and an absorption onset of ≈1010 nm.
Organic tandem solar cell with PBDB-TF:IT-4F as active layer for the front cell and PTB7-Th:IEICO-4Cl:PC71BM as active layer for the real cell gained a power conversion efficiency (PCE) of 15.26%.
High efficient non-fullerene acceptor
with highly conjugated core
Expanded absorption range to near infrared
Large dipole moment induced by chloride substituents
Worldwide shipping
Quick and reliable shipping
High purity
>99% High purity
Device structure: ITO/TIPD/PBDB-TF:IT-4F/m-PEDOT/TIPD/PTB7-Th:IEICO-4Cl:PC71BM/MoO3/Ag
| Thickness (nm) | VOC (V) | JSC (mA cm-2) | FF (%) | PCE (%) |
| 105/150 nm | 1.58 | 14.36 | 67.27 | 15.26 |
Our high purity (>99%) IEICO-4Cl is available for priority dispatch (lead times may apply for large quantities) and ships free with qualifying orders.
General Information
| CAS Number | 2240998-88-1 |
| Chemical Formula | C114H114Cl4N4O4S4 |
| Purity | >99% (1HNMR) |
| Full Name | 2,2′-((2Z,2′Z)-(((4,4,9-tris(4-hexylphenyl)-9-(4-pentylphenyl)-4,9-dihydro-s-indaceno[1,2-b:5,6-b dithiophene-2,7-diyl)bis(4-((2-ethylhexyl)oxy)thiophene 5,2-diyl))bis(methanylylidene))bis(5,6-dichloro-3-oxo-2,3-dihydro-1H-indene-2,1-diylidene))dimalononitrile |
| Molecular Weight | 1874.22 g/mol |
| Absorption | λmax 802 nm (in chloroform), 887 nm (in film), NIR absorption |
| HOMO / LUMO | HOMO = -5.56 eV, LUMO = -4.23 eV; Eg = 1.33 eV [1] |
| Solubility | Chloroform, chlorobenzene |
| Form | Dark blue powder/crystals |
| Synonyms | IEICO-Cl |
| Classification / Family | NFAs, n-type non-fullerene electron acceptors, organic semiconducting materials, low band-gap small molecule, small molecular acceptor, organic photovoltaics, organic photodetectors (OPDs), polymer solar cells, NF-PSCs |
Chemical Structure
Pricing
| Batch | Quantity | Price |
| M2341A1 | 100 mg | £290 |
MSDS Documentation
Literature and Reviews
-
Efficient Semitransparent Organic Solar Cells with Tunable Color enabled by an Ultralow-Bandgap Nonfullerene Acceptor, Y. Cui et al., Adv. Mater., 29 (43), 1703080 (2017); DOI: 10.1002/adma.201703080.
- Efficient Organic Tandem Solar Cells Enabled by Solution-Processed Interconnection Layer and Fine-Tuned Active Layer, R. Zeng et al., Adv. Optical Mater., 9 (21), 2101246 (2021); DOI: 10.1002/adom.202101246.
- Nonfullerene Acceptors for Semitransparent Organic Solar Cells, S. Dai et al., Adv. Energy Mater., 1800002 (2018); DOI: 10.1002/aenm.20180000.
Related Products
Semiconducting polymers for bulk heterojunction, OPV, OLED, OFET and perovskite interfaces and solar cell research.


