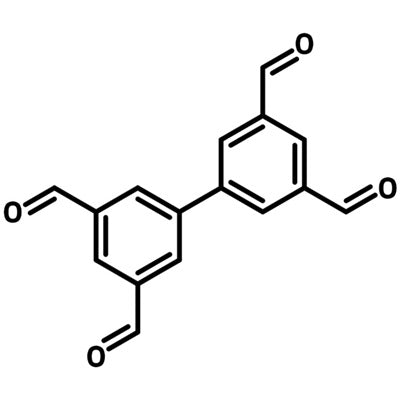
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde
CAS Number 150443-85-9
Carbaldehyde Monomers, COF Ligands, Materials, Monomers,Covalent Organic Frameworks (COFs) Biphenyl Ligand
A linear linker for COFs in applications of effective chromium adsorption and carbon dioxide reduction
Specifications | MSDS | Literature and Reviews
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde (TABP), CAS number 150443-85-9, is consisted of a biphenyl linker with four carbaldehyde groups at 3,3',5,5'-positions of the biphenyl structure.
Derived from [1,1'-biphenyl]-3,3',5,5'-tetracarbaldehyde and bearing hydroxyl groups, dual-pore COF-BTA-DHBZ showed a fast removing process of chromium (VI) with a maximum adsorption capacity up to 384 mg g−1. Study revealed that the hydroxyl groups on the backbone of the COF played a critical role for the capture of Cr (VI).
A donor-acceptor (D–A) structure of Co-COF-BT, comprising (1,1′-biphenyl)-3,3′,5,5′-tetracarbaldehyde and 4,4′-(2,1,3-benzothiadiazole-4,7-diyl)bis[benzenamine], possesses excellent photocatalytic CO2 reduction activity with a CO yield of 2423 μmol g−1h−1 and selectivity >99% under visible light. It is believed that the introduction of Co2+ to the COF pores provides additional active sites. It is the Co-S bond in Co-COF-BT that generates an additional channel for ultrafast electron migration from the COF-BT unit to the Co2+ active center to increase charge mobility and reduce the energy barrier.
MOF and COF ligands
Aldehyde ligand for cross-linked COF networks
Worldwide shipping
Quick and reliable shipping
High purity
>98% High purity
Facile reactions
Aldehyde possesses excellent reactivity
General Information
| CAS Number | 150443-85-9 |
|---|---|
| Chemical Formula | C16H10O4 |
| Full Name | [1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde |
| Molecular Weight | 266.25 g/mol |
| Synonyms | TABP, BTA, 1,1'-Biphenyl-3,3',5,5'-tetracarbaldehyde |
| Classification / Family | Biphenyls, COF ligands |
Chemical Structure
Product Details
| Purity | >98% |
|---|---|
| Melting Point | N/A |
| Appearance | White to light yellow powder/crystals |
MSDS Documentation
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde MSDS Sheet
Literature and Reviews
-
Cheap transition metal reinforced donor–acceptor covalent organic frameworks for CO2 photoreduction, L. Ai et al, Chem. Eng. J., 475, 146106 (2023); DOI: /10.1016/j.cej.2023.146106.
-
Efficient Removal of Cr(VI) from Aqueous Solutions by a Dual-Pore Covalent Organic Framework, F. Cui et al., Adv. Sustainable Syst., 3 (4), 1800150 (2019); DOI: 10.1002/adsu.201800150.
-
2D Covalent Organic Frameworks with Kagome Lattice: Synthesis and Applications, J. Tu et al., Chem, Eur. J., e202302380 (2023); DOI: 10.1002/chem.202302380.
![[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde CAS 150443-85-9](http://www.ossila.com/cdn/shop/files/bta-150443-85-9-chemical-structure-title.jpg?v=1718706710&width=380)