BDTT-Cl-Sn
CAS Number 2239295-69-1
Chemistry Building Blocks, Heterocyclic Building Blocks, Materials, Monomers,High purity BDTT-Cl-Sn, for applications in highly efficient OSCs and OPVs
For the synthesis of small molecular donors and low band gap semiconducting polymers
Specifications | MSDS | Literature and Reviews
2,6-bis(trimethytin)-4,8-bis(5-(2-ethylhexyl)-4-chlorothiophen-2-yl)benzo[1,2-b:4,5-b’]dithiophene (BDTT-Cl-Sn, CAS number 2239295-69-1) is an electron-rich building block used for the synthesis of either small molecular donors or low band-gap polymer semiconductors (like PBDB-T-2Cl) for highly-efficient OSCs and OPV applications.
It is believed that electron density can be delocalized more effectively to the halogen atom in Th-Cl than in Th-F. Also bearing two ethylhexyl side chains, with fused thiophenes and thiophenes as electron-rich pendants, BDTT-Cl-Sn offers good solubility for the targeted materials.
General Information
| CAS Number | 2239295-69-1 |
| Chemical Formula | C40H56Cl2S4Sn2 |
| Molecular Weight | 973.46 g/mol |
| Full Name | 2,6-bis(trimethytin)-4,8-bis(5-(2-ethylhexyl)-4-chlorothiophen-2-yl)benzo[1,2-b:4,5-b’]dithiophene |
| Synonyms |
|
| Classification / Family | Thiophene, Fused thiophene, Benzo-dithiophene heterocylic aromatics, Five-membered ring, Semiconductor synthesis intermediates, Low band-gap polymers, OFETs, Organic photovoltaics, Polymer solar cells |
Chemical Structure
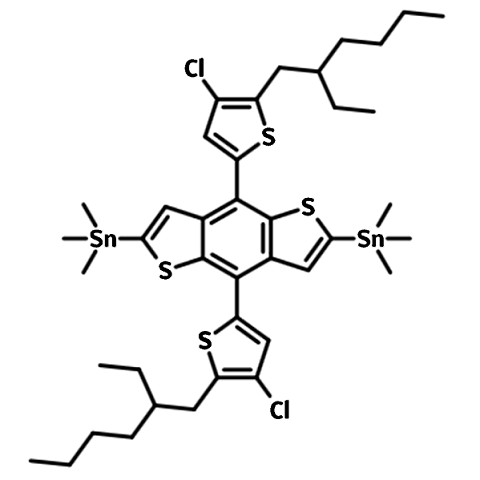
Product Details
| Purity | >98% (1H-NMR) |
| Melting Point | N/A |
| Appearance | Light yellow crystals |
MSDS Documentation
Literature and Reviews
- Over 14% Efficiency in Polymer Solar Cells Enabled by a Chlorinated Polymer Donor, S. Zhang et al., Adv.Mater., 30, 1800868 (2018); DOI: 10.1002/adma.201800868.
- Chlorine substituted 2D-conjugated polymer for high-performance polymer solar cells with 13.1% efficiency via toluene processing, Q. Fan et al., Nano Energy 48, 413–420 (2018); doi: 10.1016/j.nanoen.2018.04.002.
- Fluorination vs. chlorination: a case study on high performance organic photovoltaic materials, Y. Zhang et al., Sci. China Chem., 61 (10), 1328–1337 (2018); doi: 10.1007/s11426-018-9260-2.
