PCDTBT
CAS Number 958261-50-2
Interface Polymers, OFET & OLED Polymer Materials, Hole Transport Layer Materials, Luminosyn™ Polymers,Next Generation Donor Polymer for OPVs
A stable material which produces better efficiencies
Overview | Specifications | MSDS | Literature and Reviews | Technical Support
PCDTBT (CAS 958261-50-2) is one of the next generation donor materials developed for organic photovoltaics to produce better efficiencies and lifetimes. The key properties of PCDTBT result from the lower HOMO/LUMO levels which lead to advantages over standard organic photovoltaic materials of increased open circuit voltage, longer wavelength absorption, and improved stability under ambient conditions.
Power conversion efficiencies of up to 6.7% have been achieved in our own labs using PCDTBT in a standard reference architecture using PEDOT:PSS as a hole interface and calcium/aluminum as an electron interface. By using advanced interface materials and antireflection coatings PCDTBT has also achieved up to 7.2% in the literature [1].
PCDTBT from Ossila was used in the high-impact paper (IF 29.37), All-Inkjet-Printed, All-Air-Processed Solar Cells, S. Jung et al., Adv. Energy Mater., 1400432 (2014); DOI: 10.1002/aenm.201400432.
The lower lying HOMO level of PCDTBT makes it much more stable under ambient conditions and therefore an ideal candidate to use with large area deposition methods such as ink-jet printing, spray coating, and blade coating. However, for these deposition techniques, uniform, aggregate free coatings are essential and so lower molecular weights are often desirable.
Contact us for large orders of 5-10 grams, available with a lead time of 4-6 weeks.
The Luminosyn™ Range
General Information
| CAS Number | 958261-50-2 |
|---|---|
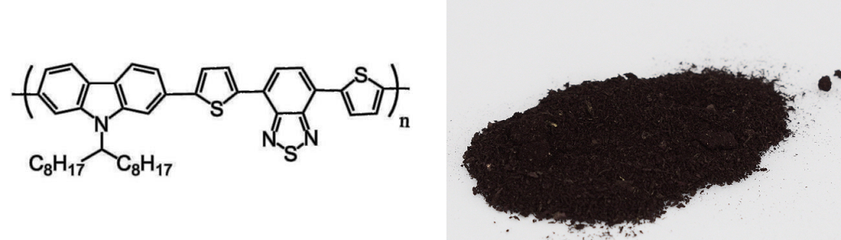
| Chemical Formula | (C43H47N3S3)n |
| Full Name | Poly[N-9'-heptadecanyl-2,7-carbazole-alt-5,5-(4',7'-di-2-thienyl-2',1',3'-benzothiadiazole)] |
| Molecular Weight | See Batch Details for information |
| HOMO / LUMO | HOMO = -5.4 eV, LUMO = -3.6 eV |
| Soluble In | Chloroform, chlorobenzene, dichlorobenzene and trichlorobenzene |
| Recommended Processing Solvents at 10mg/ml | Chloroform, chlorobenzene |
| Synonyms | PCDTBT |
| Classification or Family | Polycarbazoles, Heterocyclic five-membered ring, Organic semiconducting materials, Low band gap polymers, Organic photovoltaics, Polymer solar cells, OLEDs, OFETs and Perovskite solar cells |
Chemical Structure
Usage Datasheet
For high performance organic photovoltaics with efficiencies of 6% and above poly[N-9'-heptadecanyl-2,7-carbazole-alt-5,5-(4',7'-di-2-thienyl-2',1',3'-benzothiadiazole)] (PCDTBT). We have achieved efficiencies of 6.7% in our own labs using a standard reference architecture of PEDOT:PSS as a hole interface and calcium/aluminum as an electron interface (see below for fabrication details). Our paper published in Nature Scientific Reports titled Molecular weight dependent vertical composition profiles of PCDTBT:PC71BM blends for organic photovoltaics explores the effect and optimization of molecular weight.
JV curve from PCDTBT in a standard reference device
PCDTBT UV-Vis absorption spectrum
PCDTBT photoluminescence spectrum
Solution Details
Ossila’s reference devices were made by dissolving PCDTBT at 4 mg/ml in anhydrous chlorobenzene using a stir-bar and hotplate at 80°C overnight. This was then mixed with Ossila’s dry 95%/5% C70-PCBM powder in a 1:4 blend ratio to produce an overall concentration of 20 mg/ml. The blend solution was heated with a stir-bar on a hotplate at 80 °C for 2 hours before cooling to room temperature over 10 minutes and filtering with a 0.45 μm PTFE filter immediately prior to spinning at 700 rpm to give a film of approx. 70 nm.
Device Structure
Glass / ITO / PEDOT:PSS AI 4083 / PCDTBT:PC70BM / Ca / Al
Ossila’s pre-patterned ITO substrates (S211) with 100 nm (20 Ω/square) ITO were cleaned with the following procedure:
- 5 minutes sonication in hot 1% Hellmanex III
- 2 x hot dump rinses, 1x cold dump rinse
- 5 minutes sonication in warm IPA
- 3 x cold dump rinses
- 5 minutes sonication in hot 10% NaOH solution
- 2 x cold dump rinses then stored in DI water until use
- N2 blow dry before spin-coating the hole transport layer (no further cleaning or surface treatment required)
PEDOT:PSS AI 4083 was filtered through a 0.45 µm PES filter (C2009S1) before spin coating at 6000 rpm in air to produce a layer 30 nm thick. The coated substrates were then stored on a hotplate at 150 °C before transfer into a glove box and a further bake of 150 °C for 10 mins to remove any residual moisture. The active ink was spin cast and the cathode strip wiped clean using chlorobenzene before transfer to an evaporator where 2.5 nm of Ca followed by 100 nm of Al were deposited at <10-6 mbar. The substrates were then annealed at 80 °C for 15 mins on a hotplate in the glove box before protecting with the Ossila encapsulation system. Measurement was performed under ambient conditions using a Newport 92251A AM1.5 100 mW/cm2 solar simulator and NREL certified silicon reference cell.
MSDS Documentation
Batch Details
In general, PCDTBT is used at lower concentrations than P3HT (typically 4 to 7 mg/ml) and higher blend ratios (1:4 PCDTBT:PC70BM) and as such 100 mg of PCDTBT will make around 500 devices on Ossila's standard ITO substrates (20 x 15 mm) even assuming 50% material loss in filtration and solution preparation. Please note that as the higher molecular weight fractions have a lower yield we are now operating differential pricing policy. See below for more details on separation, yield and differential pricing.
| Batch | Mw | Mn | PDI | Stock Info |
|---|---|---|---|---|
| M1311 | 34,900 | 16,200 | 2.15 | Low stock |
| M0131A1 | 42,661 | 18,804 | 2.27 | In stock |
| M0131A2 | 10,145 | 5,319 | 1.90 | In stock |
| M0131A3 | 33,063 | 12,240 | 2.70 | In stock |
Literature and References
Please note that Ossila has no formal connection to any other authors or institutions in these references.
- Efficient, Air-Stable Bulk Heterojunction Polymer Solar Cells Using MoOx as the Anode Interfacial Layer, Y. Sun et al., Advanced Materials, 23, 2226-2230 (2011)
- Optimizing the efficiency of carbazole co-polymer solar-cells by control over the metal cathode electrode, D.C. Watters et al., Organic Electronics, 13, 1401-1408 (2012)
