Germanium Disulfide (GeS2) Powder and Crystals
CAS Number 12025-34-2
2D Materials, Anode Active Materials, Battery Materials, Inorganic Electronic Materials,Low price, high purity 2D metal germanium disulfide powder and crystal
For the development of next-generation electronics, optoelectronics, and nanotechnology
Technical Data | MSDS | Structure | Literature and Reviews | Related Products | Resources | Technical Support
Germanium disulfide (GeS2), CAS number 12025-34-2, belongs to the group IV layered transition metal dichalcogenides (TMDCs). A promising anisotropic semiconductor with unique vdW structure, germanium disulfide is currently of particular research interest into its potential in energy storage applications such as solid state batteries.
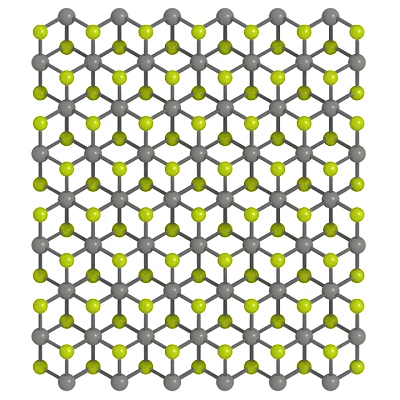
Unlike SnS2 or SnSe2 which has a simple 1T structure with the central Sn atoms are octahedrally coordinated by six outer S/Se atoms, GeS2 has more complex structure. GeS2 has a layered structure with tetrahedral coordination with complex structural patterns, containing 48 atoms in the bulk unit cell (space group P21/c). In these structures, each layer is composed of eight distorted, edge- and corner-sharing tetrahedral [GeS4 ] motifs in one unit cell.
High Purity
High purity Germanium Disulfide ≥99.995% powder
Worldwide Shipping
Quick and reliable shipping
Low Price
Low price Germanium Disulfide
Different Forms
Available in powder and crystals
Nanoflakes of 2D GeS2 with a thickness of 4.2 nm achieved via mechanical exfoliation is expected to show strong in-plane anisotropy in electrical, optical, and mechanical properties and has been used as polarization-sensitive photo-detectors in the short wave region.
We supply low price germanium disulfide in several different forms for a range of applications.

Germanium Disulfide Powder
Can be used for preparation of germanium disulfide nanoplates and ultrathin films
Sold by weight
≥99.995% purity
From £220
Germanium Disulfide by Size
Can be used to produce single or few-layer germanium disulfide sheets via mechanical or liquid exfoliation
Small (≥10 mm2) or medium (≥25 mm2) crystals available*
≥99.999% purity
From £680
*Typical representative size, areas/dimensions may vary
Bulk single germanium disulfide crystal is most commonly used as sources from which single or few-layer sheets can be obtained via either mechanical or liquid exfoliation.
Germanium disulfide powder can also be used to prepare GeS2 nanosheets and nanoparticles by liquid-exfoliation (normally assisted by sonication).
Technical Data
| CAS Number | 12025-34-2 |
| Chemical Formula | GeS2 |
| Molecular Weight | 136.77 g/mol |
| Bandgap | 2.265 eV (monoclinic); 2.599 eV (Tetragonal) |
| Preparation | Synthetic - Chemical Vapor Transport (CVT) |
| Structure | Tetragonal |
| Electronic Properties | 2D semiconductor |
| Melting Point | 825 °C (lit.) |
| Color | Black |
| Synonyms | Germanium(IV) sulfide |
| Classification / Family | Transition metal dichalcogenides (TMDCs), 2D semiconductor materials, Nano-electronics, Nano-photonics, Photovoltaic, Materials science |
Product Details
| Form | Purity |
|---|---|
| Powder | ≥99.995% |
| Crystal | ≥99.999% |
Pricing Table
| Product Code | Form | Size/Weight* | Price |
|---|---|---|---|
| M2305C1 | Powder | 500 mg | £220 |
| M2305C1 | Powder | 1 g | £350 |
| M2305A10 | Crystal | Small (≥10 mm2) | £680 ea. |
| M2305A25 | Crystal | Medium (≥25 mm2) | £1200 ea. |
*Typical representative size, areas/dimensions may vary
Shipping is free for qualifying orders.
MSDS Documents
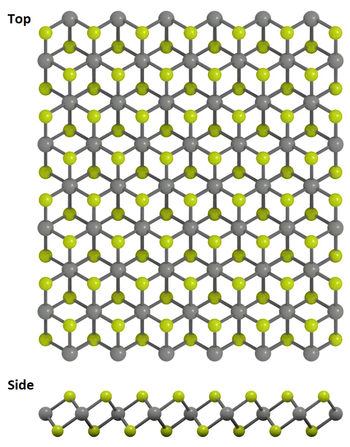
Structure of Germanium Disulfide
GeS2 crystallizes in the tetragonal P4_2/nmc space group. The structure is two-dimensional and consists of two GeS2 sheets oriented in the (0, 0, 1) direction. Ge4+ is bonded to four equivalent S2- atoms to form corner-sharing GeS4 tetrahedra. All Ge–S bond lengths are 2.27 Å. S2- is bonded in a water-like geometry to two equivalent Ge4+ atoms.
Tetragonal GeS2 is considered a structure of high symmetry: a GeS2 monolayer in which all tetrahedra are connected via corners, resulting in a much more ordered arrangement. This structure corresponds to a single slab of the high-pressure, layer-structured, tetragonal HgI2-type phase.
Literature and Reviews
- Highly reversible and superior Li-storage characteristics of layered GeS2 and its amorphous composites, G-K. Sung et al., CS Appl. Mater. Interfaces, 8, 43, 29543–29550 (2016); DOI: 10.1021/acsami.6b10994.
- Sub-Angstrom Characterization of the Structural Origin for High In-Plane Anisotropy in 2D GeS2, X. Wang et al., ACS Nano, 14, 4456−4462 (2020); DOI: 10.1021/acsnano.9b10057.
- Establishing the structure of GeS2 at high pressures and temperatures: a combined approach using x-ray and neutron diffraction, A. Zeidler et al., J. Phys.: Condens. Matter 21 , 474217 (2009); DOI: 10.1088/0953-8984/21/47/474217.
Related Products
We stock a wide range of 2D materials available to purchase online. Please contact us if you cannot find what you are looking for.



