Exciplex and Excimer: Formation and Emission

An exciplex (or excited complex) is a complex formed from two different molecules, where one molecule is in an excited state (donor) and the other is in the ground state (acceptor). Both are typically conjugated semiconducting molecules. This short-lived complex will occur when the molecules are in close proximity to one another. The two molecules in this state have a lower energy than if the two existed separately in their excited and ground states.
What makes exciplexes unique is that the two molecules would not form a complex if both were in the ground state. They can therefore only form if the monomers interact before the excited one has time to relax. If the two monomers are the same species, the complex is instead called an excimer (excited dimer).
Exciplexes are useful as TADF materials due to their small triplet-singlet energy difference. Electrons in the triplet state can therefore be thermally transferred to the singlet state from which they can fluoresce. This makes them promising for use in high efficiency third-generation OLEDs. Additionally, exciplexes are commonly used in phosphorescent (second-generation) OLEDs and also have potential as emitters in white light OLEDs. Research is ongoing into how these materials can improve device efficiencies and lifetimes, particularly for blue OLEDs.
Fundamentals of Exciplex Formation
When one molecule is in an excited state, it is possible for two conjugated semiconducting molecules in close proximity to form an excited dimer complex. When the two molecules are the same species, the complex is called an excimer (excited dimer); when they are different, the complex is known as an exciplex (excited complex).
This process can be described by:
Here, A and B are monomers and * denotes an excited state. In the case of an excimer, A=B, whereas for an exciplex, A≠B.
When both molecules are in the ground state, electronic interactions between them are not possible as all orbitals are completely full. Therefore, the molecules are repulsive due to Coulomb interactions. However, when one of them is in the excited state, interactions with the orbitals of the ground state molecule stabilize the excited state molecule, leading to a lower energy state as well as an attraction between the molecules. This is usually facilitated by π-π stacking, the mutual attraction between aromatic rings.
Exciplex Components
Exciplexes are formed via charge transfer (CT) interactions between a donor and an acceptor in the excited state. Upon excitation, an electron is transferred from the highest occupied molecular orbital (HOMO) of the donor to the lowest unoccupied molecular orbital (LUMO) of the acceptor. This charge-separated state is stabilized by weak non-covalent interactions, resulting in low singlet-triplet energy gaps.
The choice of donor and acceptor plays a critical role in exciplex performance. Ideal donors and acceptors should exhibit complementary electronic properties to ensure effective CT interactions. To gain a high quantum efficiency, exciplex systems of the donor and acceptor should meet certain conditions:
- The difference in energy level of the donor and the acceptor molecules should be enough to ensure effective formation of intermolecular excited states. It is suggested that stable exciplex could be formed only if the HOMO and the LUMO energy levels of donor are different from those of the acceptor by more than 0.4 eV.
- To avoid the efficiency drop caused by the reverse energy transfer of excitons, donor and acceptor molecules should have higher ET than that of the formed exciplex.
- The ET values of the donor and acceptor molecules must closely match or reach the singlet energy level of the formed exciplex, as the ET of the exciplex is approximately equal to its singlet energy level.
Common donors include carbazole derivatives, phenothiazine, and triphenylamine, while popular acceptors encompass triazines, benzothiadiazoles, and quinoxalines. Balancing the HOMO-LUMO energy levels of the donor-acceptor pair is essential to achieving a small ∆EST and efficient TADF. Computational modeling and experimental optimization are used to choose or design materials with desired electronic properties. Strategies such as incorporating heteroatoms and conjugated moieties can modulate the molecular orbitals to achieve an ideal energy gap.
Importance of Spatial Proximity
The spatial arrangement of donor and acceptor molecules significantly impacts exciplex formation and TADF efficiency. Proximity and favorable alignment facilitate stronger CT interactions. Non-covalent interactions such as hydrogen bonding, π-π stacking, and van der Waals forces can enhance molecular interaction and stability, optimizing the exciplex’s photophysical properties.
The host matrix in which the exciplex is embedded affects its emission characteristics and stability. High triplet energy hosts are often employed to prevent exciton quenching. Furthermore, the polarity of the host matrix can influence the CT state energy, enabling emission color tuning. Minimizing the energy offset between the S1 and T1 states requires precise control over the donor and acceptor energy levels.
The concentration of donor and acceptor molecules affects exciplex formation and emission. Optimal doping levels ensure efficient CT interactions while minimizing aggregation-induced quenching or phase separation.
Exciplex Emission
As the excimer/exciplex has a lower energy than the molecule, when it relaxes back to the ground state, the photon emitted is of lower energy than the emission of the monomer. Hence, it is redshifted. In addition to this redshift, the excimer/exciplex emission is also broad and featureless (no transitions to vibrational states). This is because the exciplex ground state is very unstable and the two ground state monomers are repulsive, giving a continuum of emission energies. The diagram below illustrates how the energy of the system varies with the separation of the monomers A and B.
In the above diagram, you can see that when the separation between A and B* is sufficiently small, the total energy of the system is lower. Any emission from this state will therefore be redshifted compared to the monomer emission. In addition to this, the emission is broader due to the unstable nature (no energy well) of the ground-state excimer, and featureless due to the lack of excimer vibrational states.
Excimer/exciplex formation depends heavily on the density of the molecules. If the density is too low, the excited state will relax back down to its ground state before encountering another monomer. Therefore, exciplex emission will increase as the density is increased and emission from the monomer will be reduced. Excimers/exciplexes are generally formed in solutions due to the high likelihood of collisions between monomers. Although collisions are not possible in the solid state, in the crystalline form stacking can occur. This leads to excimers when one molecule is excited. Similarly, in amorphous solids, molecules that are in close proximity to one another can result in excimers.
Exciplexes as Emitting Materials in OLEDs
The emission wavelength of exciplexes can be tuned by varying the donor-acceptor pair, making them versatile for OLED applications. However, exciplexes generally do not tend to be as efficient as the high-efficiency emitters already in use due to high non-radiative rates. However, lots of exciplexes have a very small singlet-triplet excited state energy gap, ΔST, an essential property for thermally-activated delayed fluorescence (TADF).
In TADF materials, ΔST is limited by the electron exchange energy: if this is large, so is ΔST. In exciplexes, however, the exchange energy is much smaller as the electron and hole are located on different molecules. ΔST is therefore much smaller than in standard TADF materials. Resultingly, reverse intersystem crossing (reverse ISC) in which an excited triplet electron can be transferred to the excited singlet state through thermal energy (provided it exceeds ΔST) becomes highly likely. TADF exciplexes, therefore, are highly fluorescent and can have theoretical efficiencies of up to 100%.
Due to this broad spectrum, excimers and exciplexes are also promising as emitters in broadband OLEDs and white OLEDs (WOLEDs). For example, if the monomer chosen emits in the blue and has a broad excimer emission in the red and green, a white OLED can be produced with relative ease. This is advantageous as one layer can be used to generate all required colors. It is difficult, however, to exactly tailor the emission spectrum as it is highly-sensitive to the density of the monomers, their relative abundance, and their emission spectra.
Exciplexes as Host Materials in OLEDs
Exciplexes are also highly promising as host materials in phosphorescent (2nd generation) OLEDs. The function of host materials is to disperse the emitting material in order to reduce density-dependent emission quenching (a form of fluorescence quenching). Because exciplexes consist of two different monomers, there is an asymmetric charge density in the system and therefore charge transfer can occur between the molecules. This leads to one molecule acting as a donor and one as an acceptor. Exciplex-forming co-hosts are therefore useful for balancing charge mobilities. Furthermore, with their low driving voltages, higher external quantum efficiency (EQE), and long device lifetimes, they have been found to be an improvement on previous host materials.
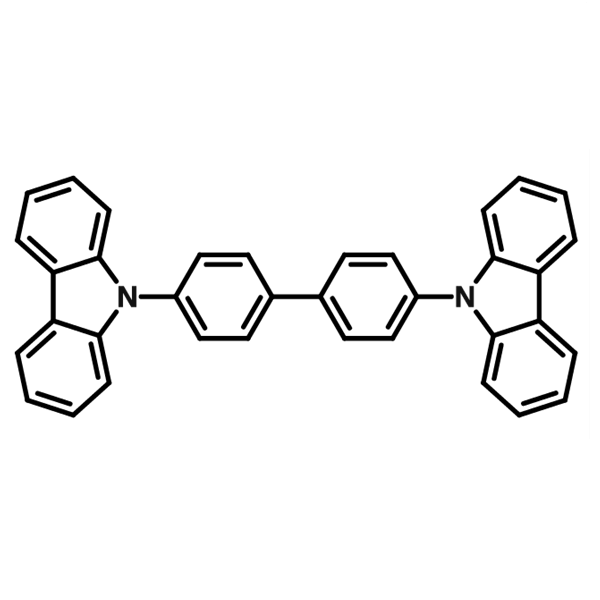
Usually in an exciplex co-host, one material is p-type (hole transport) and the other is n-type (electron transport). This allows for a better carrier injection and transport compared to single host devices and will also result in higher efficiencies. As the electrons and holes are able to recombine over the whole emitter layer, the exciton density is lower and less localized, thus, triplet-triplet annihilation is reduced. Additionally, the lifetimes of OLEDs that use exciplex co-hosts are improved because of the separate electron and hole transport materials. In single hosts, the transport of both types of carrier will lead to degradation and reduced lifetimes. An example of a charge transfer exciplex as host materials is TCTA and TPBi:
Exciplex co-hosts are now the host of choice for red and green phosphorescent OLEDs; however, blue LEDs still remain problematic due to the high triplet energies required to facilitate energy transfer. This is because Intersystem Crossing (ISC) is more likely to occur if the singlet and triplet states are at similar energies. The hunt for blue OLED materials is therefore ongoing.
TADF Materials
Learn More
Spontaneous and Stimulated Emission are essential phenomena to consider when evaluating the suitability of organic semiconducting molecules for laser applications. Amplified spontaneous emission (ASE) serves as a reliable indicator of its potential. ASE is the process used by lasers to amplify light. There are three important optical transitions that happen during ASE:
Read more... TADF Exciplex OLED Technology
TADF Exciplex OLED Technology
A thermally activated delayed fluorescence (TADF) exciplex is an excited-state species that can exhibit thermally activated delayed fluorescence or transfer its energy to a lower-energy emitter.
Read more...Further Reading
- Sarma, M. (2018). Exciplex: An Intermolecular Charge-Transfer Approach for TADF. ACS Appl. Mater. Interfaces 10, 23, DOI: 10.1021/acsami.7b18318
Contributing Authors
Written by
PhD Student Collaborator
Edited by
Application Scientist

