L8-BO-F, highly efficient non-fullerene acceptor
Higher solubility and morphology than L8-BO, giving impressive device performances
Specifications | Pricing and Options | MSDS | Literature and Reviews
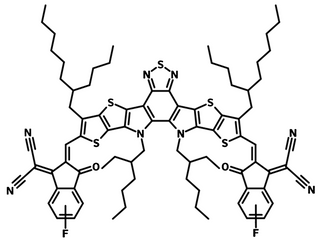
Structurally, L8-BO-F has the same thienothienopyrrolo-thienothienoindole (TTP-TTI) core units as L8-BO (L8-BO-2F) with mono-fluorinated peripheral 2-(5 or 6-fluoro-3-oxo-2,3-dihydro-1H-inden-1-ylidene)malononitrile end-groups.
L8-BO-F is another highly efficient non-fullerene acceptor that belongs to Y6 family. Similar to L8-BO, it has a branched 2-butyloctyl side chain which not only promotes solubility but also improves the morphology to achieve better packed film with higher intermolecular stackings, balanced charge transport and to reduce charge combination. L8-BO-F is also believed to suppresses the non-radiative recombination leading to a reduce the voltage loss.
High efficient non-fullerene acceptor
With highly conjugated core
Improved film quality
Owing to the branched butyloctyl side chain
Worldwide shipping
Quick and reliable shipping
High purity
>99% pure
Outstanding device performance with PCE of 18.66% has achieved while a binary BTP-eC9 and L8-BO-F non-fullerene acceptors were used with polymer donor PM6. The polymer donor and mixed NFAs give complementary absorptions in the visible and NIR region with well matched energy alignment.
Device structure: ITO/PEDOT:PSS AI 4083/PM6:BTP-eC9:L8-BO-F (1:1.2)/PNDIT-F3N/Ag
| Thickness (nm) | VOC (V) | JSC (mA cm-2) | FF (%) | PCE (%) |
| 100 | 0.853 | 27.35 | 80.00 | 18.66 |
General Information
| CAS Number | N/A |
| Chemical Formula | C84H92F2N8O2S5 |
| Purity | >99% (1H NMR) |
| Full Name | 2,2'-((2Z,2'Z)-((12,13-bis(2-ethylhexyl)-3,9-(2-butyloctyl)-12,13-dihydro-[1,2,5]thiadiazolo[3,4-e]thieno[2",3’':4’,5']thieno[2',3':4,5]pyrrolo[3,2-g]thieno[2',3':4,5]thieno[3,2-b]indole-2,10-diyl)bis(methanylylidene))bis(5 or 6-fluoro-3-oxo-2,3-dihydro-1H-indene-2,1-diylidene))dimalononitrile |
| Molecular Weight | 1444.00 g/mol |
| HOMO / LUMO | HOMO = -5.59 eV, LUMO = -3.85 eV [1] |
| Solubility | Chloroform, chlorobenzene and dichlorobenzene |
| Form | Dark blue powder/crystal |
| Synonyms | L8-BO-F |
| Classification / Family | BTP series NFAs, n-type non-fullerene electron acceptors, Organic semiconducting materials, Low band-gap small molecule, Small molecular acceptor, Organic photovoltaics, Polymer solar cells, NF-PSCs. |
Chemical Structure
MSDS Documentation
Pricing
| Batch | Quantity | Price |
| M2306A1 | 50 mg | £240 |
| M2306A1 | 100 mg | £380 |
| M2306A1 | 250 mg | £780 |
*for 5 - 10 grams order quantity, the lead time is 4 - 6 weeks
Literature and Reviews
- A Well-Mixed Phase Formed by Two Compatible Non-Fullerene Acceptors Enables Ternary Organic Solar Cells with Efficiency over 18.6%, Y. Cai et al., Adv. Mater., 33 (33); 2101733 (2021); DOI: 10.1002/adma.202101733.
-
A facile strategy for third-component selection in non-fullerene acceptor-based ternary organic solar cells, Y. Li et al., Energy Environ. Sci., 14, 5009-5016 (2021); DOI: 10.1039/D1EE01864G.
- Non-fullerene acceptors with branched side chains and improved molecular packing to exceed 18% efficiency in organic solar cells, C. Li et al., Nat. Energy, 6, 605–613 (2021); DOI: 10.1038/s41560-021-00820-x.
Related Products
Semiconducting polymers for bulk heterojunction, OPV, OLED, OFET and perovskite interfaces and solar cell research.

 L8-BO-F MSDS sheet
L8-BO-F MSDS sheet
