Rhenium Disulfide (ReS2) Powder and Crystal
CAS Number 12038-63-0
2D Materials, Low Dimensional Materials, Materials, Transition Metal Chalcogenides (TMCs),Low price, high purity 2D metal rhenium disulfide powder and crystals
For the development of next-generation electronics, optoelectronics, and nanotechnology
Technical Data | MSDS | Structure | Literature and Reviews | Related Products | Resources and Support
Rhenium disulfide (ReS2, CAS number 12038-63-0) is a 2D layered semiconductor. Unlike most 2D layered materials, ReS2 behaves as a stack of electronically and vibrationally decoupled monolayers, even in the bulk form. Raman spectrum and photoluminescence properties are independent of the number of layers. This suggests that ReS2 could offer a novel system to study mesoscopic physics of 2D systems without the limitation of obtaining large-area, monolayer-thick flakes.
High Purity
≥99.999% rhenium diselenide Crystal
Worldwide Shipping
Quick and reliable shipping
Powder & Crystal
Different Forms of Rhenium Disulfide
Sheets
Produce single or few-layer sheets
Rhenium disulfide is a direct band gap semiconductor that has exhibited outstanding optoelectronic performance due to its weak interlayer coupling. Compared to black phosphorus (BP), ReS2 is a naturally-occurring yet extremely rare TMDC so it is environmentally stable which makes it an ideal candidate for applications in ambient conditions. ReS2 has been found to be a diamagnetic semiconductor, an n-type photovoltaic material with a 1.4 eV optical gap, and exhibits bi-axial optical and electrical properties.
We supply low price rhenium disulfide in several different forms for a range of applications.

Rhenium Disulfide Powder
Can be used for preparation of rhenium disulfide nanoplates and ultrathin films
Sold by weight
≥99.995% purity
From £220

Rhenium Disulfide Crystals by Size
Can be used to produce single or few-layer rhenium disulfide sheets via mechanical or liquid exfoliation
Small (≥10 mm2) or medium (≥25 mm2) crystals available*
≥99.999% purity
From £520
*Typical representative size, areas/dimensions may vary
Bulk single rhenium disulfide crystal is most commonly used as sources from which single or few-layer sheets can be obtained via either mechanical or liquid exfoliation.
Rhenium disulfide powder can also be used to prepare ReS2 nanosheets and nanoparticles by liquid-exfoliation (normally assisted by sonication), especially when it is the case of foreign elements such as lithium or sodium cations being inserted between layers by the process of intercalation. Liquid exfoliation can provide mass production of such products.
Technical Data
| CAS Number | 12038-63-0 |
| Chemical Formula | ReS2 |
| Molecular Weight | 250.34 g/mol |
| Bandgap | ~1.35 eV (direct) |
| Preparation | Synthetic - Chemical Vapor Transport (CVT) |
| Structure | Triclinic |
| Electronic Properties | 2D semiconductor |
| Melting Point | N/A |
| Color | Black |
| Synonyms | Rhenium sulfide, Bis(sulfanylidene)rhenium |
| Classification / Family | Transition metal dichalcogenides (TMDCs), 2D semiconductor materials, Nano-electronics, Nano-photonics, Photovoltaic, Materials science |
Product Details
| Form | Purity |
|---|---|
| Powder | ≥99.995% |
| Crystal | ≥99.999% |
Pricing Table
| Product Code | Form | Size/Weight* | Price |
|---|---|---|---|
| M2139C1 | Powder | 500 mg | £220 |
| M2139C1 | Powder | 1 g | £350 |
| M2139A10 | Crystal | Small (≥10 mm2) | £520 ea. |
| M2139A25 | Crystal | Medium (≥25 mm2) | £850 ea. |
*Typical representative size, areas/dimensions may vary
Shipping is free for qualifying orders.
MSDS Documents
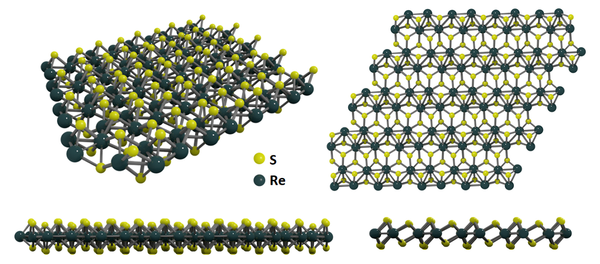
Structure of Rhenium Disulfide
ReS2 unit cell is derived from hexagonal symmetry towards triclinic - a distorted 1T structure. An ReS2 single layer is comprised of three atomic layers, 'S–Re–S', where Re and S are joined by covalent bonds and each rhenium is coordinated to six sulfur atoms in approximately octahedral geometry. Each sulfur atom is bonded to three Re atoms. Each Re atom also groups into parallelograms of four Re atoms, hence offering opportunities to introduce built-in planar anisotropy into composite heterostructures.
Like other 2D layered materials, the adjacent layers in ReS2 are also coupled by weak van der Waals (vdW) forces to form bulk crystals.
Unlike most of the TMDCs, ReS2 has an unique structure with its in-plane 1D chains of Re atoms. These are arranged into linked parallelograms - forming Re4 “diamond” shapes along the b axis. Such a structure makes it an optically bi-axial material which shows exceptionally anisotropic electrical and optical behavior for linearly-polarized light.
Literature and Reviews
- Synthesis and Characterization of ReS2 and ReSe2 Layered Chalcogenide Single Crystals, B. Jariwala et al., Chem. Mater., 28, 3352−3359 (2016); DOI: 10.1021/acs.chemmater.6b00364.
- Rhenium Dichalcogenides: Layered Semiconductors with Two Vertical Orientations, L. Hart et al., Nano Lett., 16, 1381−1386 (2016); DOI: 10.1021/acs.nanolett.5b04838.
- Growth of two-dimensional rhenium disulfide (ReS2) nanosheets with a few layers at low temperature, S. Kim et al., CrystEngComm, 19, 5341 (2017); DOI: 10.1039/c7ce00926g.
Related Products
We stock a wide range of 2D materials available to purchase online. Please contact us if you cannot find what you are looking for.



