Specifications | MSDS | Literature and Reviews | Technical Support
PDPP3T (CAS number 1198291-01-8) is used as active layer material for OPV devices with 6.71% PCE reported when a solvent mixture of DCB/Chloroform/DIO was used.
Being electron rich, PDPP3T is also used as an effective spiro-OMeTAD replacement hole-transport layer material for perovskite solar cells [1], as well as in organic field-effect transistors [2, 3] and printed electronics.
Please enquire for 5 - 10 grams order quantity, the lead time is 4-6 weeks.
The Luminosyn™ Range
General Information
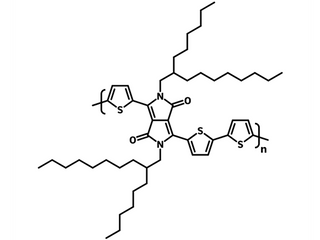
| Full name | Poly{2,2′-[(2,5-bis(2-hexyldecyl)-3,6-dioxo-2,3,5,6- tetrahydropyrrolo[3,4-c ]pyrrole-1,4-diyl)dithiophene]- 5,5′-diyl-alt-thiophen-2,5-diyl} |
| Synonyms | PDPP3T, DPP3T |
| Chemical formula | (C50H72N2O2S3)n |
| CAS number | 1198291-01-8 |
| HOMO / LUMO | HOMO = -5.17 eV, LUMO = -3.61 eV [1] |
| Solubility | Toluene, chloroform, chlorobenzene, dichlorobenzene |
| Recommended Processing Solvents at 10mg/ml | Chloroform |
| Classification / Family | Pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione, Heterocyclic five-membered ring, Organic semiconducting materials, Low band gap polymers, Organic Photovoltaics, Polymer Solar Cells, OFETs |
Batch Details
| Batch number | MW | Mn | PDI | Stock info |
|---|---|---|---|---|
| M0991A3 | 80,129 | 41,303 | 1.94 | In stock |
Chemical Structure
MSDS Documentation
Literature and Reviews
- Solution processed pristine PDPP3T polymer as hole transport layer for efficient perovskite solar cells with slower degradation, A. Dubey et al., Sol. Energ. Mat. Sol. Cells 145, 193–199 (2016);DOI: 10.1016/j.solmat.2015.10.008.
- Optimization of charge carrier transport balance for performance improvement of PDPP3T-based polymer solar cells prepared using a hot solution, J. Wang et al., Phys. Chem. Chem. Phys. 17, 9835--9840 (2015); DOI: 10.1039/c5cp00963d.
- Effects of 2,3,5,6-tetrafluoro-7,7,8,8-tetracyanoquinodimethane doping on diketopyrrolopyrrole-based, low crystalline, high mobility polymeric semiconductor, S. Yoon et al., Appl. Phys. Lett. 107, 133302 (2015); doi: 10.1063/1.4932530.
