PDIN, ETL material used to modify and enhance surface morphology
High purity and available online with free worldwide shipping on qualifying orders
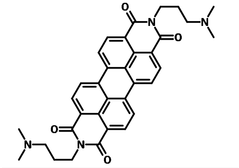
N,N'-Bis[3-(dimethylamino)propyl]perylene-3,4,9,10-tetracarboxylic diimide (PDIN), CAS number 117901-97-0, is alcohol soluble with the assistance of acetic acid (2% in volume) and a traditional cathode interface material (CIM), similar to that of PFN.
PDIN can be used as electron transporting layer (ETL) material to effectively modify and enhance surface morphology, resulting in a favorable out-of-plane π–π stacking with associated lamellar orientation. PDIN can also be used as a surface modifier on ZnO when ZnO is used as ETL material for organic electronic devices.
PDIN is not soluble in chlorinated solvents, such as chlorobenzene and chloroform. By using trifluoroethanol as solvent for PDIN in perovskite solar cells, high quality PDIN thin film can be formed onto the perovskite layer and the erosion problem can be overcome by using conventional alcohol solvents such as methanol and ethanol.
PDIN, also known as DAPER DNA precipitation reagent, has a reputation of precipitating DNAs at extremely low concentrations.
Cathode interlayer molecule
For high efficient OPV applications
Improved surface morphology
Favourable out-of-plane π–π stacking
Worldwide shipping
Quick and reliable shipping
High purity
>98% pure
General Information
| Full name | N,N'-Bis[3-(dimethylamino)propyl]perylene-3,4,9,10-tetracarboxylic diimide |
| Purity | 98% (1H NMR) |
| Synonyms |
|
| Chemical formula | C34H32N4O4 |
| Molecular weight | 560.65 g/mol |
| CAS number | 117901-97-0 |
| HOMO / LUMO | HOMO = -6.05 eV LUMO = -3.72 eV [1] |
| Melting point (DSC) | > 300 °C |
| Form | Red powder/crystals |
| Absorption | λmax 480 nm (film) |
| Solubility/processing solvents | Methanol, ethanol, trifluoroethanol |
| Classification / Family | Perylene diimide (PDI), Organic semiconducting materials, Organic Photovoltaics, OLED, Cathode interlayer materials (CIMs) |
Chemical Structure
MSDS Documentation
Pricing
| Batch | Quantity | Price |
| M2232A1 | 1 g | £280 |
| M2232A1 | 5 g | £1100 |
| M2232A1 | 10 g | £2000 |
Literature and Reviews
- Perylene diimides: a thickness-insensitive cathode interlayer for high performance polymer solar cells, Z. Zhang et al., Environ. Sci., 7, 1966 (2014); DOI: 10.1039/c4ee00022f.
- N,N'-bis[3,3'-(dimethylamino)propylamine]-3,4,9, 10-perylenetetracarboxylic Diimide, a Dicationic Perylene Dye for Rapid Precipitation and Quantitation of Trace Amounts of DNA, Z. Liu et al., Anal. Biochem, 5; 236(1):139-45 (1996); DOI: 10.1006/abio.1996.0142.
- A non-fullerene small molecule processed with green solvent as an electron transporting material for high efficiency p-i-n perovskite solar cells, J. Miao et al., Org. Electonics, 52, 200-205 (2018); DOI: 10.1016/j.orgel.2017.10.028.
