Formamidinium Iodide (FAI)
CAS Number 879643-71-7
Perovskite Precursor Materials, Perovskite Materials, MaterialsFormamidinium iodide, precursor for optimum solar conversion efficiencies
High quality, low price, and available to buy for fast, secure dispatch
Specifications | MSDS | Pricing and Options | Literature and Reviews | Resources and Support
Formamidinium iodide (CAS number 879643-71-7) is an iodide salt of formamidine, the simplest amidine. Formamidinium iodide is often used as a precusor for synthesizing formamidinium lead iodide applied in perovskite solar cells. Formamidinium lead iodide shows a narrower bandgap than the commonly used methylammonium lead iodide (1.48 eV compared to ~1.57 eV), and hence lies closer to that favorable for optimum solar conversion efficiencies. With an approach of FAPbI3 crystallization by the direct intramolecular exchange of dimethylsulfoxide (DMSO) molecules intercalated in PbI2 with formamidinium iodide, device with performance over 20% has been fabricated.
Perovskite precursor material
for the synthesis of organic-inorganic hybrid perovskite
Worldwide shipping
Quick and reliable shipping
Used in multiple electronic applications
including FETs, LEDs, and PVs
High purity
> 99.9% non-recrystallized and recrystallized grade available
Formamidinium Iodide (FAI) from Ossila was used in the high-impact paper (IF 9.229), Using Soft Polymer Template Engineering of Mesoporous TiO2 Scaffolds to Increase Perovskite Grain Size and Solar Cell Efficiency, Q. Lian et al., ACS Appl. Mater. Interfaces 12, 18578–18589 (2020); DOI: 10.1021/acsami.0c02248.
Specifications
| CAS number | 879643-71-7 |
|---|---|
| Chemical formula | CH5IN2 |
| Molecular weight | 171.97 g/mol |
| Synonyms | FAI, Formamidine hydroiodide |
| Classification / Family | Perovskite precursor materials, Perovskite solar cells, OLEDs |
Product Details
| Purity |
M552: 99.995% (ICP-OES) M551: >99.995% (further purified by double recrystallization from 99.9% grade in ethanol) |
|---|---|
| Melting point | 242 °C |
| Color | White powder/crystals |
Chemical Structure
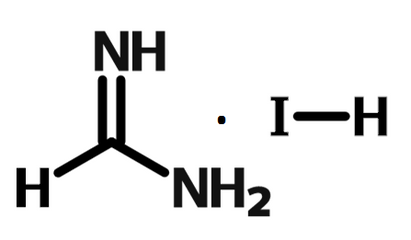
| Device structure | FTO/TiO2/FAPbIBr2/spiro-OMeTAD/Au [1] |
|---|---|
| JSC (mA cm-2) | 23.3 |
| VOC (V) | 0.94 |
| FF (%) | 65 |
| PCE (%) | 14.2 |
| Device structure | FTO/TiO2/(FAPbI3)0.85(MAPbBr3)0.15/PTAA/Au [4] |
|---|---|
| JSC (mA cm-2) | 22.5 |
| VOC (V) | 1.11 |
| FF (%) | 73.2 |
| PCE (%) | 18.4 |
| Device structure | FTO/bl-TiO2/mp-TiO2/FAPbI3 (DMSO)/PTAA/Au [5] |
|---|---|
| JSC (mA cm-2) | 24.7 |
| VOC (V) | 1.06 |
| FF (%) | 77.5 |
| PCE (%) | 20.2 |
Pricing
| Grade | Order Code | Quantity | Price |
|---|---|---|---|
| 99.995% purity | M552 | 5 g | £130 |
| 99.995% purity | M552 | 10 g | £220 |
| 99.995% purity | M552 | 25 g | £300 |
| >99.995% purity | M551 | 5 g | £200 |
| >99.995% purity | M551 | 10 g | £330 |
MSDS Documentation
Formamidinium iodide MSDS sheet
Applications
Formamidinium lead iodide shows a narrower bandgap than the commonly used methylammonium lead iodide (1.48 eV compared to ~1.57 eV), and hence lies closer to that favorable for optimum solar conversion efficiencies [1]. Spin-coating the formamidinium iodide (FAI) plus PbI2 precursor solution in N,N-dimethylformamide (DMF) initially resulted in discontinuous perovskite films. However, by adding a small amount of hydroiodic acid (HI) to the stoichiometric FAI, extremely uniform and continuous films were formed.
Controlled humidity is another deciding factor that affects the film morphology, crystallinity, and optical and electrical properties of FAPbI3 [2]. 16.6% PCE was achieved with low relative humidity of 2%, with the device efficiency dropped to about half (8.6%) when the humidity was 40%.
Low-volatility additives such as FACl and MACl are good candidates for assisting in the crystallization of phase pure α-FAPbI3 via the formation of intermediate mixtures, which prohibits the crystallization of the δ-FAPbI3 phase [3]. It also has been observed that the black perovskite-type polymorph (α-phase), which is stable at relatively high temperatures (above 160 oC), turned into the yellow FAPbI3 polymorph (δ-phase) in an ambient humid atmosphere. Results show that incorporation of MAPbBr3 into FAPbI3 stabilizes the perovskite phase of FAPbI3 and improves the power conversion efficiency of the solar cell to more than 18% under a standard illumination of 100 mW/cm2 [4].
With an approach of FAPbI3 crystallization by the direct intramolecular exchange of dimethylsulfoxide (DMSO) molecules intercalated in PbI2 with formamidinium iodide, device with performance over 20% has been fabricated [5].
Literature and Reviews
- Formamidinium lead trihalide: a broadly tunable perovskite for efficient planar heterojunction solar cells, G. E. Eperon et al., Energy Environ. Sci., 7, 982-988 (2014), DOI: 10.1039/C3EE43822H.
- Controlled Humidity Study on the Formation of Higher Efficiency Formamidinium Lead Triiodide-Based Solar Cells, S. Wozny et al., Chem. Mater., 27 (13), 4814–4820 (2015), DOI: 10.1021/acs.chemmater.5b01691.
- Additive-Modulated Evolution of HC(NH2)2PbI3 Black Polymorph for Mesoscopic Perovskite Solar Cells, Z. Wang et al., Chem. Mater., 27 (20), 7149–7155 (2015), DOI: 10.1021/acs.chemmater.5b03169.
