1,3,6,8-Tetrakis(4-formylphenyl)pyrene
CAS Number 1415238-25-3
Carbaldehyde Monomers, Chemistry Building Blocks, COF Ligands, Materials,Covalent Organic Frameworks (COFs) Pyrene Ligand
An aldehyde functionalized bridging ligand linker for COFs in application of photocatalytic water splitting, gas uptake and energy storage
Specifications | MSDS | Literature and Reviews
1,3,6,8-Tetrakis(4-formylphenyl)pyrene (TFPPY), CAS number 1415238-25-3, tecton has a pyrene core with four side 4-formylphenyl functional groups which grows the structure via condensations reactions.
Constructed from 1,3,6,8-tetrakis(4-formylphenyl)pyrene and 4,4'-(5,6-dichlorobenzo[c][1,2,5]thiadiazole-4,7-diyl)dianiline as the building blocks, two-dimensional (2D) covalent organic framework, namely Py-ClTP-BT-COF, showed great solar-to-hydrogen energy conversion efficiency with photocatalytic H2 evolution rates of 177.50 μmol h−1 and a high apparent quantum efficiency (AQE) of 8.45%.
With the introduction of oxygen atoms into the framework to increase the number of photogenerated electrons, pyrene-based amide COF, TFPPy–DP, demonstrated photocatalytic reduction and removal of toxic uranyl under visible light with 82% removal percentage of 238 ppm uranyl at pH 3 in 2 h of irradiation.
Microporous carbon material, Py-PDT POP-600, obtained by the carbonization of Py-PDT POP with 6,6′-(1,4-phenylene)bis(1,3,5-triazine-2,4-diamine) (PDA-4NH2) and 1,3,6,8-tetrakis(4-formylphenyl)pyrene repeating units, showed a high nitrogen content with high surface area of 314 m2 g−1, high pore volume, and good thermal stability. Py-PDT POP-600 demonstrated excellent performance in CO2 uptake of 2.7 mmol g−1 at 298 K and a high specific capacitance of 550 F g−1 at 0.5 A g−1.
MOF and COF ligands
Aldehyde ligand for cross-linked COF networks
Worldwide shipping
Quick and reliable shipping
High purity
>97% High purity
Facile reactions
Aldehyde possesses excellent reactivity
General Information
| CAS Number | 1415238-25-3 |
|---|---|
| Chemical Formula | C44H26O4 |
| Full Name | 1,3,6,8-Tetrakis(4-formylphenyl)pyrene |
| Molecular Weight | 618.67 g/mol |
| Synonyms | TFPPy, 4,4',4'',4'''-(pyrene-1,3,6,8-tetrayl)tetrabenzaldehyde |
| Classification / Family | Pyrenes, COF ligands |
Chemical Structure
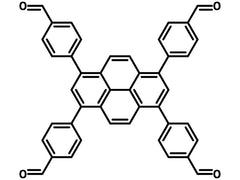
(TFPPY) Chemical Structure, 1415238-25-3.
Product Details
| Purity | >97% |
|---|---|
| Melting Point | N/A |
| Appearance | Yellow to orange powder/crystals |
MSDS Documentation
1,3,6,8-Tetrakis(4-formylphenyl)pyrene MSDS Sheet
Literature and Reviews
-
Modulating Benzothiadiazole-Based Covalent Organic Frameworks via Halogenation for Enhanced Photocatalytic Water Splitting, W. Chen et al., Angew. Chem. Int. Ed., 132 (39), 17050-17057 (2020); DOI: 10.1002/ange.202006925.
-
Amide Linkages in Pyrene-Based Covalent Organic Frameworks toward Efficient Photocatalytic Reduction of Uranyl, J. Kang et al., ACS Appl. Mater. Interfaces, 14 (51), 57225–57234 (2022); DOI: 10.1021/acsami.2c16702.
-
Carbonized Aminal-Linked Porous Organic Polymers Containing Pyrene and Triazine Units for Gas Uptake and Energy Storage, A. Mousa et al., Polymers, 15(8), 1891 (2023); DOI: 10.3390/polym15081891.
