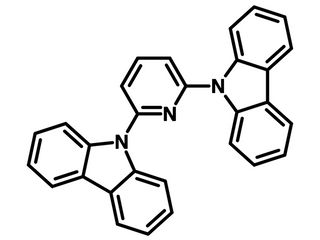
PYD-2Cz (PYD2)
CAS Number 168127-49-9
Fluorescent Host Materials, High Purity Sublimed Materials, Host Materials, Materials,PYD-2Cz, bipolar host material for PhOLEDs
Ideal candidate for hosting blue electro-phosphorescence OLEDs, 2,6-Bis(9H-carbazol-9-yl)pyridine, CAS No. 168127-49-9, Sublimed ≥99.0%
PYD-2Cz is composed of two electron-donating carbazole substituents with an electron deficient pyridine core unit. PYD-2Cz is a commonly-used bipolar host material for phosphorescent organic light‐emitting diodes (PHOLEDs). This is due to its wide band-gap and ability to be both an electron acceptor and donor.
With higher triplet energy levels (T1 = 2.93 eV), PYD-2Cz is an ideal candidate for hosting blue electro-phosphorescence OLEDs.
General Information
| CAS number | 168127-49-9 |
|---|---|
| Full name | 2,6-Bis(9H-carbazol-9-yl)pyridine |
| Chemical formula | C29H19N3 |
| Molecular weight | 409.48 g/mol |
| Absorption | λmax 241 nm, 290 nm in DCM |
| Fluorescence | λem 373 nm in THF |
| HOMO/LUMO | HOMO = 5.7 eV, LUMO = 2.2 eV; T1 = 2.93 eV [1] |
| Synonyms | PYD2, PYD-2, mCPy |
| Classification / Family | Carbazole, Bipolar host materials, TADF green emitter materials, Phosphorescent organic light-emitting devices (PHOLEDs), Sublimed materials |
Product Details
| Purity | Sublimed >99.0% (HPLC) |
|---|---|
| Melting point | 202°C (lit.)TGA: >250 °C (0.5% weight loss) |
| Appearance | Off-white powder/crystals |
*Sublimation is a technique used to obtain ultra pure-grade chemicals. For more details about sublimation, please refer to the Sublimed Materials.
Chemical Structure
Device Structure(s)
| Device structure | ITO/TAPC (40 nm)/PYD-2Cz:2wt% of Ir(MDQ)2acac (30 nm)/BmPyPhB (40 nm)/LiF (0.8 nm)/Al (150 nm) [2] |
|---|---|
| Color | Red |
| Max. Current Efficiency | 15.0 cd/A |
| Max. EQE | 8.5% |
| Max. Power Efficiency | 12.8 Im/W |
| Device structure | ITO (100 nm)/PEDOT:PSS (35 nm)/PLEXCORE UT-314 (20 nm)/PYD2:Cu(I)-iBuPyrPHOS 3:1 (30 nm)/3TPYMB(70 nm)/LiF (1 nm)/Al [3] |
|---|---|
| Color | Green |
| Max. Current Efficiency | 65.4 cd/A |
| Max. EQE | 21% |
| Device structure | ITO (130 nm)/PEDOT:PSS (30 nm)/PLEXCORE UT-314(45 nm)/PYD2:Cu(I)-complex 7:3 (27 nm)/3TPYMB (50 nm)/LiF (2 nm)/Al (100nm) [4] |
|---|---|
| Color | Green |
| Max. Current Efficiency | 73 cd/A |
| Max. EQE | 23% |
| Device structure | ITO/PEDOT:PSS/ PYD2: 4 wt% Au (III) complex 5 (60 nm)/ TPBi/LiF (1.2 nm)/Al (100 nm) [5] |
|---|---|
| Color | Sky-blue |
| Max. Current Efficiency | 70.4 cd/A |
| Max. EQE | 23.8% |
| Max. Power Efficiency | 47.3 lm/W |
| Max. Luminance | 33,470 cd/m2 |
MSDS Documentation
Literature and Reviews
- Analyzing Bipolar Carrier Transport Characteristics of Diarylamino-Substituted Heterocyclic Compounds in Organic Light-Emitting Diodes by Probing Electroluminescence Spectra, K. Son et al., Chem. Mater., 20, 4439–4446 (2008); DIO: 10.1021/cm8004985.
- Phenanthro[9,10-d]imidazole based new host materials for efficient red phosphorescent OLEDs, D. Tavgeniene et al., Dyes and Pigments, 137, 615-621 (2017); DIO: 10.1016/j.dyepig.2016.11.003.
- Highly Efficient Organic Light-Emitting Diode Using A Low Refractive Index Electron Transport Layer, A. Salehi et al., Adv. Optical Mater., 1700197 (2017); DOI: 10.1002/adom.201700197.