PDCBT, highly efficient fullerene free polymer solar cells
High quality and high purity semiconducting polymer available online for priority dispatch
Specifications | MSDS | Literature and Reviews | Technical Support
PDCBT (CAS number 1609536-17-5) is a medium-to-wide bandgap polymer semiconductor that belongs to the polythiophene family. With two electron-withdrawing carboxylate side groups, PDCBT is relatively electron deficient when it is compared with P3HT. PDCBT has a main absorption peak (in film) located at 551 nm, 37 nm red-shifted compared to that of P3HT.
PDCBT has been used in highly efficient fullerene free polymer solar cells. These have achieved device power conversion efficiency of 10.16% with ITIC used as the non-fullerene acceptor [6]. Ta-WOx doped PDCBT has been reported as efficient low-loss hole transport layer in hybrid organohalide lead perovskite solar cell not only to enhance the device performance achieving maximum efficiencies of 21.2% but also offering more than 1000 hours of light stability with almost 95% of its efficiency maintained [7]
The Luminosyn™ Range
General Information
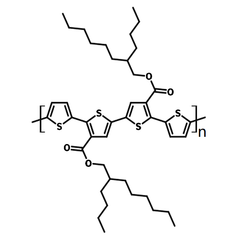
| Full name | Poly[2,2''''-bis[[(2-butyloctyl)oxy]carbonyl][2,2':5',2'':5'',2'''-quaterthiophene] -5,5'''-diyl] |
| Synonyms | PDCBT |
| Chemical formula | (C42H56O4S4)n |
| CAS number | 1609536-17-5 |
| HOMO / LUMO | HOMO = -5.26 eV, LUMO = -3.0 eV [1] |
| Soluble in | Chloroform, chlorobenzene and dichlorobenzene |
| Recommended Processing Solvents at 10mg/ml | Chloroform |
| Absorption* | λmax 551 nm (in film) [2] |
| Form | Brown reddish flakes/powders |
| Melting point | Tm = 282 °C (lit.) |
| Classification / Family | Polythiophenes, Organic semiconducting materials, Medium-to- wide bandgap polymers, Organic photovoltaics, Polymer solar cells, Perovskite solar cells, Hole-transport layer materials, NF-PSCs, All-polymer solar cells (all-PSCs). |
Batch Details
| Batch | Mw | Mn | PDI | Stock Info |
| M2233A1 | 101,375 | 40,659 | 2.49 | In stock |
Chemical Structure
Device Structure
Device structure: ITO/C60-SAM/perovskite/PDCBT/Ta-WOx/Au
| Thickness (nm) | VOC (V) | JSC (mA cm-2) | FF (%) | PCE (%) |
| 650 |
1.17 | 22.7 | 80 | 21.2 |
MSDS Documentation
Literature and Reviews
- Visible Sensitization for Non-Fullerene Polymer Solar Cells Using a Wide Bandgap Polymer, Y. Wang et al., J. Photopolym. Sci. Technol., 31 (2), 177-181 (2018); doi: 10.2494/photopolymer.31.177.
- A polythiophene derivative with superior properties for practical application in polymer solar cells, M. Zhang et al., Adv. Mater., 26(33), 5880-5885 (2014); DOI: 10.1002/adma.201401494.
- Thermal behavior of dicarboxylic ester bithiophene polymers exhibiting a high open-circuit voltage, R. Heuvel et al., J. Mater. Chem. C, 6, 3731--3742 (2018); DOI: 10.1039/c7tc04322h.
