Methylammonium Bromide (MABr)
CAS Number 6876-37-5
Perovskite Precursor Materials, Perovskite Materials, MaterialsMethylammonium bromide for the synthesis of perovskites
High purity precursor available to buy online (or request a quote) for fast, secure dispatch
Specifications | MSDS | Literature and Reviews | Resources and Support
Methylammonium bromide (MABr) is a precursor for the synthesis of organic-inorganic hybrid perovskites for use in FETs, LEDs and PVs.
Perovskite precursor material
for the synthesis of organic-inorganic hybrid perovskite
Worldwide shipping
Quick and reliable shipping
Used in multiple electronic applications
including FETs, LEDs, and PVs
High purity
> 99.9% non-recrystallized and recrystallized grade available
Methylammonium Bromide (MABr) from Ossila was used in the high-impact paper (IF 9.229), Using Soft Polymer Template Engineering of Mesoporous TiO2 Scaffolds to Increase Perovskite Grain Size and Solar Cell Efficiency, Q. Lian et al., ACS Appl. Mater. Interfaces 12, 18578–18589 (2020); DOI: 10.1021/acsami.0c02248.
General Information
| CAS number | 6876-37-5 |
| Chemical formula | CH6BrN |
| Molecular weight | 111.97 g/mol |
| Synonyms |
|
| HOMO / LUMO | n.a. |
| Classification / Family | Organic photovoltaics, Light-emitting diodes, Perovskite precursor materials |
Product Details
| Purity |
99.995% (M572) >99.995% (M571 further purified by recrystallization of M572) |
| Melting point | 296 °C |
| Appearance | White crystals/powder |
Chemical Structure
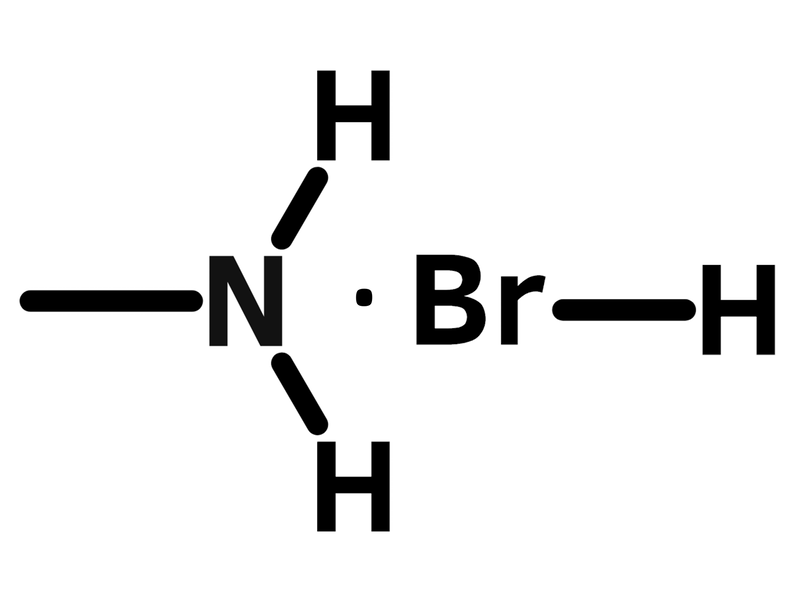
Applications
Methylammonium bromide (MABr) is a precursor of MAPbBr3 perovskites. Having a band gap of 2.3 eV (HOMO 5.68 eV, LUMO 3.38 eV) [1], MAPbBr3 perovskites have been used to tune the band gap of mixed MAPbX3 (where X is the halide I, Br and/or Cl mixtures) [2,3,4,5,6]. For this reason, bromide MAPbBr3 perovskites can be utilized as light absorbers for high-energy photons, and can serve as the front cell in tandem cells. This perovskite can provide a higher open-circuit voltage in perovskite solar cells than the iodide analog.
High-efficiency solar cells, with a VOC of up to 1.40 V, a fill factor (FF) of 79%, and a PCE of 6.7% have been reported for pure MAPbBr3 perovskite solar cells [1].
It has also been demonstrated that MAPbBr3 nanoplatelets can be employed in light-emitting diodes, exhibiting bright photoluminescence (PL) at 529 nm, with a narrow spectral band and a quantum yield up to 85% [7].
| Device structure | FTO/TiO2/(FAPbI3)0.85(MAPbBr3)0.15/PTAA/Au [8] |
| Jsc (mA cm-2) | 23.3 |
| Voc (V) | 0.94 |
| FF (%) | 65 |
| PCE | 14.2 |
MSDS Documentation
Methylammonium bromide MSDS sheet
Pricing
| Grade | Order Code | Quantity | Price |
| 98% purity | M572 | 10 g | £140 |
| 98% purity | M572 | 25 g | £260 |
| >99.5% purity | M571 | 5 g | £140 |
| >99.5% purity | M571 | 10 g | £220 |
| >99.5% purity | M571 | 25 g | £420 |
Note: Looking to place a bulk order (100 g or more) Please contact us for a quote.
Literature and Reviews
- Voltage output of efficient perovskite solar cells with high open-circuit voltage and fill factor, S. Ryu et al., Energy Environ. Sci., 7, 2614–2618 (2014). DOI: 10.1039/c4ee00762j.
- Efficient Planar Perovskite Solar Cells Based on 1.8 eV Band Gap Ch2Nh2PbI2Br Nanosheets via Thermal Decomposition, Y. Zhao et al., J. Am. Chem. Soc., 136 (35), 12241–12244 (2014). DOI: 10.1021/ja5071398.
- High Open-Circuit Voltage Solar Cells Based on Organic–Inorganic Lead Bromide Perovskite, E. Edri et al., J. Phys. Chem. Lett., 4 (6), 897–902 (2013). DOI: 10.1021/jz400348q.
