Imidazolium Iodide
CAS Number 68007-08-9
Materials, Perovskite Materials, Perovskite Precursor MaterialsOverview | Specifications | MSDS | Literature and Reviews
Imidazolium iodide (ImDI) and it derivatives are normally used as electrolyte materials for dye-sensitized solar cells (DSSCs).
Recently, with the development of perovskite solar cells and light-emitting diodes (LEDs), imidazolium iodide has also been used as an additive to perovskite solutions to inhibit the growth of 3D structures. This makes the structure less sensitive to moisture, thus improving device stability and lifetime.
General Information
| CAS number | 68007-08-9 |
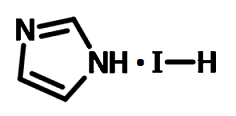
| Chemical formula | C3H5IN2 |
| Molecular weight | 195.99 g/mol |
| Synonyms | ImDI, 1H-Imidazole monohydriodide, Imidazole hydriodide, Imidazole hydroiodide |
| Classification / Family | Alkylammonium halides, Perovskite precursor materials, Perovskite solar cells, Perovskite LEDs |
Product Details
| Purity | > 99% |
| Melting point | 280 °C |
| Color | Powder/crystals |
Chemical Structure
MSDS Documentation
Literature and reviews
- Globularity-Selected Large Molecules for a New Generation of Multication Perovskites, S. Gholipour et al., Adv. Mater., 1702005 (2017); doi: 10.1002/adma.201702005.
-
Synthesis, crystal structure and phase transitions of a series of imidazolium iodides,
M. Węcławik et al., CrystEngComm, 15, 5633-5640 (2013); DOI: 10.1039/C3CE40559A.
