Imidazolium Bromide
CAS Number 101023-55-6
Perovskite Precursor Materials, Perovskite Materials, MaterialsImidazolium bromide, used as electrolytes for DSSC applications
High purity precursor available online for fast, secure dispatch
Overview | Specifications | MSDS | Literature and Reviews
Imidazolium bromide (ImDBr) is a family member of imidazolium salts that exhibits good electrical conductivity, high ionic mobility, and excellent chemical and thermal stabilities. Imidazolium bromide and its derivatives are also used as electrolytes for dye-sensitized solar cell (DSSC) applications.
Imidazolium bromide also has applications in perovskite solar cells and light-emitting diodes (LEDs).
General Information
| CAS number | 101023-55-6 |
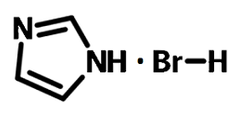
| Chemical formula | C3H5BrN2 |
| Molecular weight | 148.99 g/mol |
| Synonyms | ImDBr, 1H-Imidazole monohydrobromide, Imidazole hydrobromide |
| Classification / Family | Imidazolium halides, Perovskite precursor materials, Perovskite solar cells, Perovskite LEDs |
Product Details
| Purity | 98% |
| Melting point | 221 °C |
| Color | White - Slightly pale yellow powder/crystals |
Chemical Structure
MSDS Documentation
Imidazolium bromide MSDS sheet
Literature and reviews
- Influence of the Electrolytes on Electron Transport in Mesoporous TiO2−Electrolyte Systems, S. Kambe et al, J. Phys. Chem. B, 106 (11), 2967–2972 (2002); DOI: 10.1021/jp013397h.
- Preparation of thermally stable polymer electrolytes from imidazolium-type ionic liquid derivatives, H. Nakajima et al, Polymer, 46 (25), 11499-11504 (2005); https://doi.org/10.1016/j.polymer.2005.10.005.
