Gallium Selenide (GaSe) Powder and Crystals
Low price, high purity gallium selenide powder and crystals
Suitable for use a semiconductor, photoconductor, and in non-linear optics
Technical Data | MSDS | Structure | Literature and Reviews | Related Products | Resources and Support
Gallium selenide (GaSe), CAS number 12024-11-2, is a group XIII post-transitional mono-chalcogenide. Like transition metal dichalcogenides (TMDCs), it is a layered semiconductor. In gallium selenide, each individual layer consists of covalently-bonded stacks with sheets of Se ions on the top and bottom and two sheets of Ga ions in the middle.
High Purity
High purity gallium selenide ≥99.995% powder
Worldwide shipping
Quick and reliable shipping
Low price
Low price Gallium Selenide
Different Forms
Available in powder and crystals
As with other layered 2D structures like graphene, adjacent GaSe layers are bound by the weak van der Waals force. This makes it possible to peel the structure by mechanical or liquid exfoliation. The resultant ultra-thin few or single layer 2D gallium selenide nanosheets or nanoparticles have well-known nonlinear optical properties and a range of applications in areas including integrated optics, optical information communications and biology.
We supply high purity gallium selenide powder and crystals.

Gallium Selenide Powder
Can be used in electronic and optical devices and in the preparation of GaSe nanosheets or nanoparticles liquid chemical exfoliation or chemical vapor deposition
Available in quantities of 500 mg or 1 g
≥99.995% purity
From £310

Gallium Selenide Crystals by Size
Can be used as a semiconductor, in non-linear optics, and to produce 2D monolayer and few-layer GaSe by mechanical or liquid exfoliation
Small (>10 mm2) or medium (>25 mm2) available*
≥99.999% purity
From £520
*Typical representative size, areas/dimensions may vary
Technical Data
| CAS Number | 12024-11-2 |
| Chemical Formula | GaSe |
| Molecular Weight | 148.69 g/mol |
| Bandgap | ~2.1 eV (indirect) |
| Crystal Structure | Hexagonal |
| Electrical Properties | 2D semiconductor |
| Synonyms | Gallium(II) selenide, Selanylidenegallium |
| Classification / Family | Transition metal dichalcogenides (TMMCs), 2D semiconductor materials, Nano-electronics, Nano-photonics, Photovoltaic, Materials science |
| Preparation | Synthetic - Chemical Vapor Transport (CVT) |
| Color | Dark copper |
Product Details
| Form | Purity |
|---|---|
| Powder | ≥99.995% |
| Crystal | ≥99.999% |
*typical representative size, areas/dimensions may vary.
MSDS Documents
Structure of Bulk and 2D Gallium Selenide
The hexagonal-layered structure of gallium selenide is similar to the structure of gallium sulfide (GaS). Each individual layer consists of four sheets of atoms in the sequence 'Se-Ga-Ga-Se'.
Each Ga atom is tetragonally-coordinated to three Se atoms and one Ga atom. Strong bonding between two sheets within the individual layer is thought to be covalent in nature, with some ionic contribution.
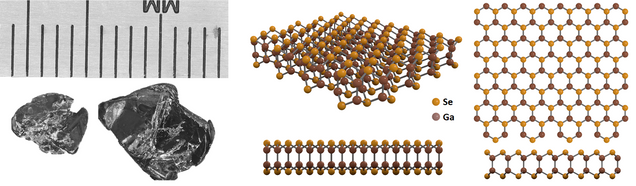
Depending on the sequence of stacking, the three most important classifications are β-GaSe, ε-GaSe, and γ-GaSe phases. Both β-GaSe and ε-GaSe are 2H hexagonal structured.
Literature and Reviews
Gallium Selenide Powder in Literature
- Layer-Dependent Nonlinear Optical Properties and Stability of NonCentrosymmetric Modification in Few-Layer GaSe Sheets, Angew. Chem. Int. Ed., 54, 1185 –1189 (2015); DOI: 10.1002/anie.201409837.
- Investigation of Second- and Third-Harmonic Generation in Few-Layer Gallium Selenide by Multiphoton Microscopy, L. Karvonen et al., Sci. Rep., 5:10334 (2015); DOI: 10.1038/srep10334.
- Highly sensitive phototransistor based on GaSe nanosheets, H. Huang et al., Appl. Phys. Lett. 107, 143112 (2015); doi: 10.1063/1.4933034.
Gallium Selenide Crystals in Literature
- Multifunctional 2D- Materials: Gallium Selenide, N. B. Singh et al., Mater. Today: Proceedings 4, 5471–5477 (2017); dio: 10.1016/j.matpr.2017.06.002.
- Effective Nonlinear GaSe Crystal Optical Properties and Applications, K. R. Allakhverdiev et al., Laser Phys., 19 (5), 1092–1104 (2009); DOI: 10.1134/S1054660X09050375.
- Copper indium gallium selenide based solar cells – a review, J. Ramanujam et al., Energy Environ. Sci., 10, 1306 (2017); DOI: 10.1039/c7ee00826k.
Related Products
We stock a wide range of 2D materials available to purchase online. Please contact us if you cannot find what you are looking for.



