COTIC-4F, NFA for responsive organic solar cells and organic photodetectors
Narrow bandgap n-type semiconductor with absorption range near infrared, available to buy online
Specifications | Pricing and Options | MSDS | Literature and Reviews
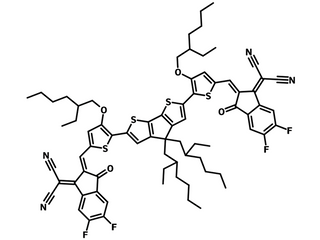
COTIC-4F has a molecular configuration of A−D'−D−D'−A that is based on an electron rich internal core of cyclopentadithiophene (CPDT) as the central donor unit flanked with alkoxythienyl units, terminated with two 2-(5,6-difluoro-3-oxo-2,3-dihydro-1H-inden-1-ylidene)malononitrile (IC-2F) acceptor units.
COTIC-4F is a narrow bandgap n-type molecular semiconductors extending its absorption from 700 nm to near infrared range (about 1100 nm) with an optical bandgap of ~1.1 eV. COTIC-4F can be used as active layer material for near infrared (NIR) responsive organic solar cells (OSCs ) and organic photodetectors (OPDs ).
High efficient non-fullerene acceptor
with highly conjugated core
Wide absorption range into near infrared region
Absorption up to 1100 nm
Worldwide shipping
Quick and reliable shipping
High purity
>99% High purity
General Information
| CAS Number | 2328118-91-6 |
| Chemical Formula | C75H78F4N4O4S4 |
| Purity | >99% (1H NMR) |
| Full Name | 2,2'-((2Z,2'Z)-(((4,4-bis(2-ethylhexyl)-4H-cyclopenta[2,1-b:3,4-b']dithiophene-2,6-diyl)bis(4-(2-ethylhexyloxy)thiophene-5,2-diyl))bis(methanylylidene))bis(5,6-difluoro-3-oxo-2,3-dihydro-1H-indene-2,1-diylidene))dimalononitrile |
| Molecular Weight | 1303.73 g/mol |
| Absorption | λmax 875 nm (in chloroform); 990 nm (Film) |
| HOMO / LUMO | HOMO = -5.26 eV, LUMO = -4.17 eV [1] |
| Solubility | Chloroform, chlorobenzene |
| Form | Dark blue powder/crystals |
| Synonyms | COTIC-4F |
| Classification / Family | NFAs, n-type non-fullerene electron acceptors, Organic semiconducting materials, Low band-gap small molecule, Small molecular acceptor, Organic photovoltaics, Polymer solar cells, NF-PSCs |
Chemical Structure
Pricing
| Batch | Quantity | Price |
| M2246A1 | 100 mg | £350 |
| M2246A1 | 250 mg | £700 |
| M2246A1 | 500 mg | £1250 |
| M2246A1 | 1 g | £2190 |
MSDS Documentation
Literature and Reviews
- Bandgap Narrowing in Non-Fullerene Acceptors: Single Atom Substitution Leads to High Optoelectronic Response Beyond 1000 nm, J. Lee et al., Adv. Energy Mater., 8 (24), 1801212 (2018); doi: 10.1002/aenm.201801212.
- Side Chain Engineering of Non Fullerene Acceptors for Near Infrared Organic Photodetectors and Photovoltaics, J. Lee et al., ACS Energy Lett., 4 (6); 1401–1409 (2019); DOI: 10.1021/acsenergylett.9b00721.
- Near-infrared organic photoelectric materials for light harvesting systems: Organic photovoltaics and organic photodiodes, B. Xie et al., InfoMat., 2, 57–91 (2020); DOI: 10.1002/inf2.12063.
Related Products
Semiconducting polymers for bulk heterojunction, OPV, OLED, OFET and perovskite interfaces and solar cell research.


