5CzBN
CAS Number 1469700-24-0
TADF Blue Dopants, Blue Dopant Materials, TADF Dopant Materials, Dopant Materials,5CzBN, light-blue TADF fluorescence emitter
High-purity (>99.0%) and available online for priority dispatch, CAS No. 1469700-24-0, 2,3,4,5,6-penta(carbazol-9-yl)benzonitrile, Penta-carbazolylbenzonitrile, TADF blue dopant
5CzBN, penta-carbazolylbenzonitrile, is sterically hindered due to the five bulky electron-donating carbazolyl groups on the benzene ring struggling for space. It is a light-blue TADF fluorescence emitter widely used for highly efficient TADF-OLED devices.
General Information
| CAS number | 1469700-24-0 |
|---|---|
| Full name | 2,3,4,5,6-penta(carbazol-9-yl)benzonitrile |
| Synonyms | 5CzCN, 2,3,4,5,6-penta(9H-carbazol-9-yl)benzonitrile |
| Chemical formula | C67H40N6 |
| Molecular weight | 929.1 g/mol |
| Absorption | λmax 325 nm, 348 nm and 420 nm in DCM |
| Fluorescence | λem 488 nm in toluene |
| HOMO/LUMO | HOMO = 5.55 eV, LUMO = 2.74 eV, T1 = 2.68 eV [1] |
| Classification / Family | Carbazole, Phthalonitrile, TADF materials, Blue dopant materials, Sublimed materials |
Product Details
| Purity | Unsublimed >98%; Sublimed >99.0% (1H NMR) |
|---|---|
| Melting point | Tg = 318 °C (lit.) |
| Appearance | Yellow powder/crystals |
*Sublimation is a technique used to obtain ultra pure-grade chemicals. For more details about sublimation, please refer to the Sublimed Materials.
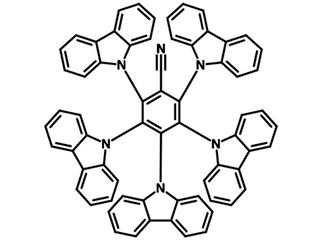
Chemical Structure
Device Structure(s)
| Device structure | ITO/HATCN (5 nm)/NPB (30 nm)/TCTA (10 nm)/mCBP:5CzBN (20 wt%) (30 nm)/DpyPA:Liq (1:1, 30 nm)/LiF (0.5 nm)/Al (150 nm) [2] |
|---|---|
| Color | Blue |
| Max. EQE | 16.7% |
| Max. Power Efficiency | 40.0 Im/W |
| Device structure | ITO (100 nm)/HATCN (10 nm)/TrisPCz (30 nm)/mCBP (10 nm)/15wt%-5CzBN:mCBP (30 nm)/T2T (10 nm)/BPyTP2 (40 nm)/LiF (0.8 nm)/Al (100 nm) [3] |
|---|---|
| Color | Blue |
| Max. EQE | 18.0% |
| Max. Power Efficiency | 49.9 Im/W |
*For chemical structure information, please refer to the cited references.
MSDS Documentation
Literature and Reviews
- Recent progress of green thermally activated delayed fluorescent emitters, Y. Im et al., J. Info. Display, 18 (3), 101-117 (2017); DOI: 10.1080/15980316.2017.1333046.
- Sterically shielded blue thermally activated delayed fluorescence emitters with improved efficiency and stability, D. Zhang et al., Mater. Horiz., 3, 145-151 (2016); doi: 10.1039/C5MH00258C.
- Excited state engineering for efficient reverse intersystem crossing, H. Noda et al., Sci. Adv., 4:eaao6910 (2018); DOI: 10.1126/sciadv.aao6910.
