DPP-DTT, PDPP2T-TT-OD
CAS Number 1260685-66-2 / 1444870-74-9
Interface Polymers, Luminosyn™ Polymers, Materials, OFET & OLED Polymer Materials, OPV Polymers, Semiconducting Polymers
DPP-DTT, high quality and high purity semiconducting polymer
High performance p-type polymer and donor material for BHJ photovoltaics
DPP-DTT is a high mobility p-type polymer, suitable for OFET and sensing and photovoltaic applications.
DPP-DTT from Ossila was used in the high-impact paper (IF 18.81), Stretchable Mesh-Patterned Organic Semiconducting Thin Films on Creased Elastomeric Substrates, S. Kim et al., Adv. Funct. Mater., 2010870 (2021); DOI: 10.1002/adfm.202010870.
Luminosyn™ DPP-DTT
Luminosyn™ DPP-DTT (also referred to as PDPP2T-TT-OD) is now available.
High molecular weight
Higher molecular weight offers higher charge mobility
High purity
DPP-DTT is purified via Soxhlet extraction with methanol, hexane and chlorobenzene under an argon atmosphere
Batch-specific GPC data
Have confidence in what you are ordering; batch-specific GPC data for your thesis or publications
Large quantity orders
Plan your experiments with confidence with polymers from the same batch
OFET and Sensing Applications
The exceptional high mobility of this polymer of up to 10 cm2/Vs [2] via solution-processed techniques, combined with its intrinsic air stability (even during annealing) has made PDPP2T-TT-OD of significant interest for OFET and sensing purposes.
While the highest mobilities require exceptional molecular weights of around 500 kD (and with commensurate solubility issues), high mobilities in the region of 1-3 cm2/Vs can still be achieved with good solution-processing at around 250 kD. As such, we have made a range of molecular weights available to allow for different processing techniques.
In our own tests, we have found that by using simple spin-coating onto an OTS-treated silicon substrate (using our prefabricated test chips), high mobilities comparable to the literature can be achieved (1-3 cm2/Vs). Further improvements may also be possible with more advanced strain-inducing deposition techniques.




Photovoltaic Applications
Although shown as a promising hole-mobility polymer for OFETs, when used as the donor material in a bulk heterojunction photovoltaic (with PC70BM as the acceptor), initial efficiencies of 1.6% were achieved for DPP-DTT [3]. The low device metrics were attributed to poor film morphology. However, a higher efficiency of 6.9% was achieved by using thicker film (220 nm) [4].
PDPP2T-TT-OD has also recently been used successfully as an active-layer dopant material in PTB7-based devices [5]. An improvement in device performance was observed, with average efficiencies increasing from 7.6% to 8.3% when the dopant concentration of DPP-DTT was 1 wt%. The use of DPP-DTT as a high-mobility hole-interface layer for perovskite hybrid devices has also been investigated [6].
Synthetic route
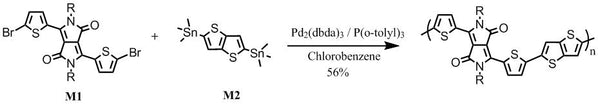
DPP-DTT synthesis: DPP-DTT was synthesised by following the procedures described in [2] and [3] (please refer to the following references):
With 2-thiophenecarbonitrile and dimethyl succinate as starting materials in t-amyl alcohol, it gave 3,6-Dithiophen-2-yl-2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione. Alkylation of 3,6-Dithiophen-2-yl-2,5-dihydropyrrolo[3,4-c]pyrrole-1,4-dione with 2-octyldodecylbromide in dimethylformamide afforded 3,6-bis(thiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione. Further bromination gave 3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione (M1).

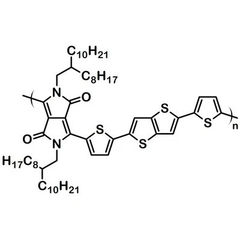
Further reaction of M1 with 2,5-bis(trimethylstannyl)thieno[3,2-b]thiophene (M2) under Stille coupling conditions gave the target polymer DPP-DTT, which was further purified via Soxhlet extraction with methanol, hexane and then chloroform.
General Information
| CAS number | 1260685-66-2 (1444870-74-9) |
| Chemical formula | (C60H88N2O2S4)n |
| HOMO / LUMO | HOMO = -5.2 eV, LUMO = -3.5 eV [2] |
| Synonyms |
|
| Solubility | o-xylene, chloroform, chlorobenzene and dichlorobenzene |
| Classification / Family | Bithiophene, Thienothiophene, Organic semiconducting materials, Low band-gap polymers, Organic photovoltaics, Polymer solar cells, OFETs |
MSDS Documentation
Pricing
| Batch | Quantity | Price |
| M0311A | 100 mg | £260 |
| M0311A | 250 mg | £520 |
| M0311A | 500 mg | £900 |
| M0311A | 1 g | £1600 |
| M0311A | 2 g | £2900 |
| M0311A | 5 g / 10 g* | Please enquire |
*For 5 - 10 grams order quantity, the lead time is 4-6 weeks.
Batch information
| Batch* | Mw | Mn | PDI | Stock info |
| M0311A4 | 152,923 | 55,143 | 2.77 | Discontinued |
| M0311A5 | 100,105 | 39,080 | 2.56 | Discontinued |
| M0311A6 | 87,278 | 37,778 |
2.31 |
Discontinued |
| M0311A7 | 111,029 | 45,803 | 2.42 | Discontinued |
| M0311A8 | 142,775 | 50,550 | 2.82 | Discontinued |
| M0311A9 | 122,960 | 42,995 | 2.86 | In Stock |
*Older batch information available on request.
References
- A High Mobility P-Type DPP-Thieno[3,2-b]thiophene Copolymer for Organic Thin-Film Transistors, Y. Li et al., Adv. Mater., 22, 4862-4866 (2010)
- A stable solution-processed polymer semiconductor with record high-mobility for printed transistors, J. Li et al., Nature Scientific Reports, 2, 754, DOI: 10.1038/srep00754 (2012)
- Synthesis of low bandgap polymer based on 3,6-dithien-2-yl-2,5-dialkylpyrrolo[3,4-c]pyrrole-1,4-dione for photovoltaic applications, G. Zhang et al., Sol. Energ. Mat. Sol. C., 95, 1168-1173 (2011)
- Efficient small bandgap polymer solar cells with high fill factors for 300 nm thick films, Li W et al., Adv Mater., 25(23):3182-3186 (2013); doi:10.1002/adma.201300017.
- Enhanced efficiency of polymer solar cells by adding a high-mobility conjugated polymer, S. Liu et al., Energy Environ. Sci., 8, 1463-1470 (2015)
- Electro-optics of perovskite solar cells, Q. Lin et al., Nature Photonics, 9, 106-112 (2015)
- A Vertical Organic Transistor Architecture for Fast Nonvolatile Memory, X. She et al., adv. Mater., 29, 1604769 (2017); DOI: 10.1002/adma.201604769.
- Solvent-Free Processable and Photo-Patternable Hybrid Gate Dielectric for Flexible Top-Gate Organic Field-Effect Transistors, J. S. Kwon et al., ACS Appl. Mater. Interfaces, 9 (6), 5366–5374 (2017); DOI: 10.1021/acsami.6b14500.
 DPP-DTT MSDS sheet
DPP-DTT MSDS sheet