Acetamidinium Iodide
CAS Number 1452099-14-7
Materials, Perovskite Materials, Perovskite Precursor MaterialsOverview | Specifications | MSDS | Literature and Reviews | Resources and Support
Like most other alkyl halides that have been used to modify perovskite structures, Acetamidinium Iodide (ADI) is often used to tune the electrical properties of perovskite solar cells and LEDs.
Compared to Formamidinium Iodide (FAI), Acetamidinium Iodide has larger-sized cations which will hinder the growth of 3D structures, thus promoting stability for perovskite electronics.
General Information
| CAS number | 1452099-14-7 |
| Chemical formula | C2H7IN2 |
| Molecular weight | 185.99 g/mol |
| Synonyms | ADI, Acetamidine Hydroiodide. 1-aminoethaniminium iodide |
| Classification / Family | Alkylammonium halides, Perovskite precursor materials, Perovskite solar cells, Perovskite LEDs |
Product Details
| Purity | 98% |
| Melting point | 100 °C |
| Colour | Powder/crystals |
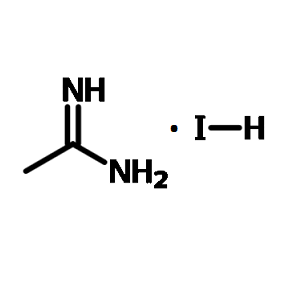
Chemical Structure
MSDS Documentation
 Acetamidinium iodide MSDS sheet
Acetamidinium iodide MSDS sheet
Literature and reviews
- Extremely efficient internal exciton dissociation through edge states in layered 2D perovskites, J.-C. Blancon et al., Science, 10.1126/science.aal4211 (2017); DOI: 10.1126/science.aal4211.
- Hybrid Germanium Iodide Perovskite Semiconductors: Active Lone Pairs, Structural Distortions, Direct and Indirect Energy Gaps, and Strong Nonlinear Optical Properties, C. C. Stoumpos et al., J. Am. Chem. Soc., 137 (21), 6804–6819 (2015); DOI: 10.1021/jacs.5b01025.
- Structure–Band Gap Relationships in Hexagonal Polytypes and Low-Dimensional Structures of Hybrid Tin Iodide Perovskites, C. C. Stoumpos et al., Inorg. Chem., 56 (1), 56–73 (2017); DOI: 10.1021/acs.inorgchem.6b02764.
