Tetrabromothiophene
CAS Number 3958-03-0
Heterocyclic Building Blocks, Monomers, Chemistry Building Blocks, MaterialsA fully brominated thiophene and a useful building block
Used for the synthesis of thiophene and fused thiophene derivatives in application of OFETs and OPVs.
Specifications | MSDS | Literature and Reviews
Tetrabromothiophene (CAS number 3958-03-0) is a fully brominated thiophene. Fully functionalized around the thiophene ring, tetrabromothiophene is used for the construction of further conjugated semiconductors as thiophene, fused thiophene or poly-fused thiophene structures in application of organic electronics, i.e. polymer solar cells and organic field-effect transistors. Fused thieno[3,2-b:4,5-b’]diindoles can be prepared with tetrabromothiophene as the starting material. Tetrabromothiophene is also an intermediate for the preparation of 3,4-dibromothiophene.
Derived from tetrabromothiophene, TPTn and TPTEn, both bearing one tetraphenylthiophene-core and either peripheral alkoxyl chains or 3,4,5-trialkoxybenzoate groups, form square, rectangular or hexagonal columnar phases depending on the mesogenic core structures and the chain length of aliphatic tails. Besides, TPTn and TPTEn luminescent liquid crystal molecules show obvious aggregation-induced emission (AIE) characteristics, the fluorescence emission intensity values measured in the aggregated state were hundred times higher than those values measured in benign solvents [1].
General Information
| CAS Number | 3958-03-0 |
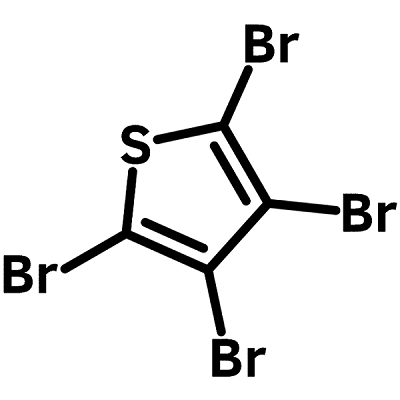
| Chemical Formula | C4Br4S |
| Full Name | Tetrabromothiophene |
| Molecular Weight | 399.72 g/mol |
| Synonyms | 2,3,4,5-tetrabromothiophene, Perbromothiophene |
| Classification / Family | Thiophenes, Semiconductor synthesis intermediates, Low band gap polymers, OLED, OFETs, organic photovoltaics |
Chemical Structure
Product Details
| Purity | >98% (1H NMR) |
| Melting point | Tm = 115 °C |
| Appearance | Off-white to orange/brown powder/crystals |
MSDS Documentation
Tetrabromothiophene MSDS Sheet
Literature and Reviews
- Luminescent liquid crystals bearing an aggregation-induced emission active tetraphenylthiophene fluorophore, L. Guo et al., J. Mater. Chem. C, 7, 4828-4837 (2019); DOI: 10.1039/C9TC00448C.
- Regioselective Palladium(0)-Catalyzed Cross-Coupling Reactions and Metal-Halide Exchange Reactions of Tetrabromothiophene: Optimization, Scope and Limitations, Đ. Tùng et al., Adv. Synth. Catal., 25 (10), 1595-1609 (2009); DOI: 10.1002/adsc.200900044.
- Photodissociation dynamics of halogenated aromatic molecules: the case of core-ionized tetrabromothiophene, L. Pihlava et al., Phys. Chem. Chem. Phys., 23, 21249-21261 (2021); DOI: 10.1039/D1CP03097C.
