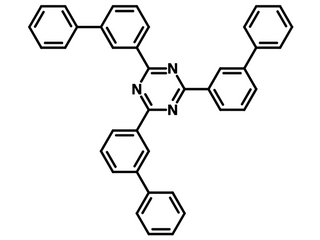
2,4,6-tris(biphenyl-3-yl)-1,3,5-triazine (T2T)
CAS Number 1201800-83-0
Electron Transport Layer Materials, Phosphorescent Host Materials, TADF Host Materials, Host Materials,T2T, host material for phosphorescent OLEDs and ETL material for TADF-OLEDs
High-purity (>99.0%) and available online for priority dispatch
2,4,6-tris(biphenyl-3-yl)-1,3,5-triazine (T2T) is a family member of triazine. It has one electron-withdrawing triazine core and three electron-rich biphenyl groups. It has been widely used as a host material for phosphorescent OLEDs and electron transport layer material (ETL) for TADF-OLEDs.
Due to its electron-deficient nature, T2T and its derivatives are also used as acceptor materials to form exciplexes in blue-emission TADF devices.
General Information
| CAS number | 1201800-83-0 |
|---|---|
| Chemical formula | C39H27N3 |
| Molecular weight | 537.65 g/mol |
| Absorption | λmax 270 nm in DCM |
| Fluorescence | λem 380 nm in DCM |
| HOMO/LUMO | HOMO = 6.5 eV, LUMO = 3.0 eV; T1 = 2.80 eV [1] |
| Synonyms | 2,4,6-tris(biphenyl-3-yl)-1,3,5-triazine |
| Classification / Family | Triazine, TADF blue emitter materials, TADF host materials, Phosphorescent organic light-emitting devices (PHOLEDs), Sublimed materials |
Product Details
| Purity | Sublimed >99.0% (HPLC) |
|---|---|
| Melting point | TGA: >300 °C (0.5% weight loss) |
| Appearance | Off-white powder/crystals |
*Sublimation is a technique used to obtain ultra pure-grade chemicals. For more details about sublimation, please refer to the Sublimed Materials.
Chemical Structure
Device Structure(s)
| Device structure | ITO/PEDOT:PSS/a-NPD (20 nm)/TCTA (5 nm)/(PPy)2Ir(acac):T2T (25 nm)/TPBi (50 nm)/LiF (0.5 nm)/Al (100 nm) [2] |
|---|---|
| Color | Green |
| Max. EQE | 17.5% |
| Max. Power Efficiency | 65.2 Im/W |
| Device structure | ITO/MoO3 (3 nm)/TAPC (25 nm)/TAPC:T2T (15 nm)/T2T (5 nm)/Bphen (30 nm)/LiF (1 nm)/Al (100 nm) [3] |
|---|---|
| Color | Green |
| Max. Current Efficiency | 40.4 cd/A |
| Max. EQE | 11.6% |
| Max. Power Efficiency | 42.2 Im/W |
| Device structure | ITO/TAPC (35 nm)/1 wt%-TBRb:25 wt%-PXZ-TRX:mCBP (30 nm)/T2T (10 nm)/Alq3 (55 nm)/LiF (0.8 nm)/Al (100 nm) [4] |
|---|---|
| Color | Yellow |
| Max. Current Efficiency | 60 cd/A |
| Max. EQE | 18.0% |
| Max. Power Efficiency | 58 Im/W |
| Device structure | ITO (100 nm)/HAT-CN (10 nm)/TAPC (30 nm)/0.65mol%-TBRb:6.3mol%-4CzIPN-Me:mCBP (30 nm)/T2T (10 nm)/Alq3 (55 nm)/LiF (0.8 nm)/Al (100 nm) [5] |
|---|---|
| Color | Green |
| Max. Current Efficiency | 73 cd/A |
| Max. EQE | 21% |
MSDS Documentation
Literature and Reviews
- Operational stability enhancement in organic light-emitting diodes with ultrathin Liq interlayers, D. Tsang et al., Sci. Rep., 6:22463 (2016); DOI: 10.1038/srep22463.
- 1,3,5-Triazine derivatives as new electron transport–type host materials forhighly efficient green phosphorescent OLEDs, H-F. Chen et al., J. Mater. Chem., 19, 8112–8118 (2009); DOI: 10.1039/b913423a.
- Thermally activated delayed-fluorescence organic light-emitting diodes based on exciplex emitter with high efficiency and low roll-off, T. Lin et al., Org. Electronics, 38, 69-73 (2016); DIO: 10.1016/j.orgel.2016.08.001.
