PNDI-T10, PCE9
CAS Number 1977539-03-9
Luminosyn™ Polymers, Materials, OFET & OLED Polymer Materials, OPV Polymers,PNDI-T10, polymer acceptor for high-efficiency all-PSCs and OFETs
High purity polymer with high electron mobility
Specifications | MSDS | Literature and Reviews | Technical Support
PNDI-T10 (also known as PCE9), CAS number 1977539-03-9, is a polymer acceptor used in high-efficiency all-polymer solar cells (all-PSCs) and organic field-effect transistors (OFETs).
PNDI-T10 is a family member of polynaphthalene diimide (which is a copolymer of naphthalene diimide with bithiophene and thiophene unit.)
All-PSCs have received great research interest in recent years. This is thanks to the blend of both electron donor and acceptor being polymers in the active layer, and how having complementary absorption covers a wider range of the solar spectrum. Using polymer acceptors (instead of fullerene) in all-PScs also has the advantages of greater morphological stability, superior mechanical properties, and higher capability of being processed with ink-jet printing techniques in flexible devices.
PNDI-T10 has been successfully used in ternary all-PSCs, achieving record performances with PCE of 9% [1]. PNDI-T10 is also a potential candidate for high performance OFET with high electron mobility.
The Luminosyn™ Range
General Information
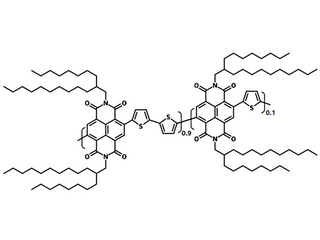
| Full name | Poly{{[N,N'-bis(2-octyldodecyl)naphthalene-1,4,5,8-bis(dicarboximide)-2,6-diyl]-alt-5,5'-(2,2'-bithiophene)}-ran-{[N,N'-bis(2-octyldodecyl)naphthalene-1,4,5,8-bis(dicarboximide)-2,6-diyl]-alt-2,5-thiophene}} |
| Synonyms | PCE9, PNDI-Th10, PNDI(2OD)-T10 |
| Chemical formula | (C62H88N2O4S2)0.9 ▪ (C58H86N2O4S)0.1 |
| CAS number | 1977539-03-9 |
| HOMO / LUMO | HOMO = -6.40 eV, LUMO = -4.10 eV [1] |
| Classification / Family | PNDI polymers, Organic n-type semiconducting materials, PNDI polymers, Organic photovoltaics, All Polymer solar cells (all-PSCs), Electron-acceptor polymers, OFETs, Perovskite solar cells. |
| Soluble in | Chloroform, chlorobenzene, dichlorobenzene |
| Recommended Processing Solvents at 10mg/ml | Chloroform |
Batch Details
| Batch | Mw | PDI | Stock Info |
| M2086A1 | 189,514 | 2.40 | Discontinued |
| M2086A2 | 103,047 | 2.08 | In stock |
Chemical Structure
MSDS Documentation
Literature and Reviews
- 9.0% power conversion efficiency from ternary all-polymer solar cells, Z. Li et al., Energy Environ. Sci., 10, 2212 (2017); DOI: 10.1039/c7ee01858d.
- High Performance All-Polymer Solar Cells by Synergistic Effects of Fine-Tuned Crystallinity and Solvent Annealing, Z. Li et al., J. Am. Chem. Soc., 138 (34), 10935–10944 (2016); DOI: 10.1021/jacs.6b04822.
- Energy-effectively printed all-polymer solar cells exceeding 8.61% efficiency, Y. Lin et al., Nano Energy 46, 428-435 (2018); https://doi.org/10.1016/j.nanoen.2018.02.035.
