DPEPO
DPEPO, one of the most popular materials used to host blue TADF-based OLEDs
High-purity and available online for priority dispatch, Bis[2-(diphenylphosphino)phenyl]ether oxide, CAS No. 808142-23-6, Sublimed ≥99.0%
DPEPO, with steric ortho-substituted diphenylphosphine oxide (DPPO) groups, is one of the most popular large band-gap materials used to host blue TADF-based OLEDs.
This substitution with electron-withdrawing DPPO moieties not only improves its thermal and morphological stability, but also makes DPEPO an electron-transport layer material. Because of its deep HOMO energy level, DPEPO also acts as a hole-blocking layer material in TADF-OLED devices.
General Information
| CAS number | 808142-23-6 |
|---|---|
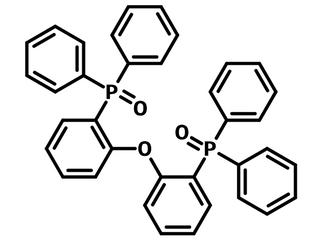
| Full name | Bis[2-(diphenylphosphino)phenyl]ether oxide |
| Synonyms | DPEPO |
| Chemical formula | C36H28O3P2 |
| Molecular weight | 570.55 g/mol |
| Absorption* | λmax 388 nm in CH2Cl2 |
| Fluorescence | λem 311 nm in CH2Cl2 |
| HOMO/LUMO | HOMO = 6.1 eV, LUMO = 2.0 eV; T1 = 3.0 eV [1] |
| Classification / Family | Diphenyl ether (DPE), TADF blue emitter host materials, Electron-transport layer materials (ETL), Hole-blocking layer materials (HBL), Phosphorescent organic light-emitting devices (PHOLEDs), Sublimed materials |
* Measurable with a USB spectrometer
Product Details
| Purity | Sublimed* >99.0% (HPLC) |
|---|---|
| Melting point | TGA: >320 °C (0.5% weight loss) |
| Appearance | White powder/crystals |
* Sublimation is a technique used to obtain ultra pure-grade chemicals, please refer to sublimed materials for more information.
Chemical Structure
Device Structure(s)
| Device structure | ITO/HATCN (5 nm)/NPB (30 nm)/TCTA (10 nm)/mCP (10 nm)/DMAC-DPS:PO-01* (0.8 wt% 30 nm)/DPEPO (10 nm)/Bphen (30 nm)/LiF (0.5 nm)/Al(150 nm) [2] |
|---|---|
| Color | White |
| Max. EQE | 20.8% |
| Max. Power Efficiency | 51.2 lm/W |
| Device structure | ITO (110 nm)/TAPC (35 nm)/mCBP (5 nm)/6 wt%-Ac-OSO:DPEPO (20 nm)/DPEPO (10 nm)/B3PyPB (40 nm)/LiF (0.8 nm)/Al (80 nm) [3] |
|---|---|
| Color | Blue |
| Max. Current Efficiency | 37.9 cd/A |
| Max. EQE | 20.5% |
| Max. Power Efficiency | 20.1 Im/W |
| Device structure | ITO/a-NPD (30 nm)/TCTA (20 nm)/CzSi (10 nm)/EML (20 nm)/ DPEPO (10 nm)/TPBI (30 nm)/LiF (0.5 nm)/Al [4] |
|---|---|
| Color | Blue |
| Max. EQE | 14.5% |
| Max. Luminesence | 2544 cd/m2 |
| Device structure | PEDOT:PSS (60 nm)/TAPC (20 nm)/mCP (10 nm)/DPEPO: TmCzTrz (25 nm)/TSPO1 (5 nm)/TPBI (20 nm)/LiF (1 nm)/Al (200 nm) [5] |
|---|---|
| Color | Blue |
| Max. EQE | 25.5% |
| Max. Power Efficiency | 52.1 Im/W |
*For chemical structure information, please refer to the cited references.
MSDS Documentation
Literature and Reviews
- Triplet exciton confinement in green organic light-emitting diodes containing luminescent charge-transfer Cu(I) complexes, Q. Zhang, et al., Adv. Funct. Mater.22, 2327–2336 (2012); DOI: 10.1002/adfm.201101907.
- Highly Efficient Simplified Single-Emitting-Layer Hybrid WOLEDs with Low Roll-off and Good Color Stability through Enhanced Förster Energy Transfer, D. Zhang et al., ACS Appl. Mater. Interfaces, 7 (51), 28693–28700 (2015); DOI: 10.1021/acsami.5b10783.
- High-Efficiency Blue Organic Light-Emitting Diodes Based on Thermally Activated Delayed Fluorescence from Phenoxaphosphine and Phenoxathiin Derivatives, S. Lee et al., Adv. Mater., 28, 4626–4631 (2016); DOI: 10.1002/adma.201506391.
