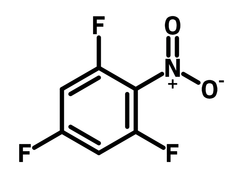
2,4,6-Trifluoronitrobenzene
CAS Number 315-14-0
Chemistry Building Blocks, Fluorinated Building Blocks, Materials, MonomersA polyfluorinated building block
Used as a synthesis intermediate for azo and azide compounds in semiconductors and photonics applications
Specifications | MSDS | Literature and Reviews
2,4,6-Trifluoronitrobenzene (CAS number 315-14-0) has three fluoro-substituents at the ortho- and para-position to the nitro group sitting at the benzene ring. 2,4,6-Trifluoronitrobenzene can be reduced to aniline, which allows further functionalization to obtain azo/azide compounds. To synthesize azo compounds, the building block is firstly converted to the diazonium salt, followed by an azo C-N coupling. Alternatively, a single-step oxidation of aniline to azo compound can be done with potassium permanganate and iron (II) sulfate as the oxidizing agents. Azide compounds can be synthesized with triflyl azide and base via diazo transfer.
The corresponding azo-compounds are used in ferroelectric devices, azo-dye liquid crystals and photoswitches (light induced trans-cis transformation of azo). The corresponding azide compounds are readily to synthesis large chromophores by 1-3 dipolar cycloaddition (click reaction) for OLEDs and dye-sensitized solar cells (DSSCs).
The fluoro-substituents enable late-stage functionalization by nucleophilic aromatic substitution to expand the functionalities of the molecules. 2,4,6-Trifluoronitrobenzene is also used for the synthesis of fluorinated polytriarylamines (F-PTAAs) which find applications as hole transporting materials in organic electronics.
Multiple functional groups
For facile synthesis
Worldwide shipping
Quick and reliable shipping
Fluorinated nitrobenzene building block
For drug discoveries, dyes, and semiconductors
High purity
>98% High purity
General Information
| CAS Number | 315-14-0 |
| Chemical Formula | C6H2F3NO2 |
| Full Name | 1,3,5-Trifluoro-2-nitrobenzene |
| Molecular Weight | 177.08 g/mol |
| Synonyms | N/A |
| Classification / Family | Fluorinated building blocks, Dyes, Nitrobenzene derivatives, Semiconductors synthesis intermediates |
Chemical Structure
Product Details
| Purity | >98% |
| Boiling Point | Tb = 183.5 °C at 760 mmHg |
| Relative Density | 1.514 g/mL at 25 °C |
| Appearance | Colorless to slightly yellow liquid |
MSDS Documentation
2,4,6-Trifluoronitrobenzene MSDS Sheet
Literature and Reviews
-
An unprecedented azobenzene-based organic single-component ferroelectric, H. Peng et al., Chem. Sci., 13, 4936–4943(2022); DOI: 10.1039/d2sc00689h.
-
Design and synthesis of visible-light-responsive azobenzene building blocks for chemical biology, J. Volarić et al., J. Org. Chem., 87, 14319−14333(2022); DOI: 10.1021/acs.joc.2c01777.
-
Light-controlled lyotropic liquid crystallinity of polyaspartates exploited as photo-switchable alignment medium, M. Hirschmann et al., J. Am. Chem. Soc., 145(6), 3615–3623(2023); DOI: 10.1021/jacs.2c12760.
