9,9′-Spirobifluorene
CAS Number 159-66-0
Chemistry Building Blocks, Materials, Monomers, Non-Heterocyclic Building BlocksHigh-Purity 9,9′-Spirobifluorene
With great thermal and chemical stabilities, spiro compounds with 9,9′-spirobifluorene core of inherent rigid structure have attracted great attention as organic functional materials.
Specifications | MSDS | Literature and Reviews
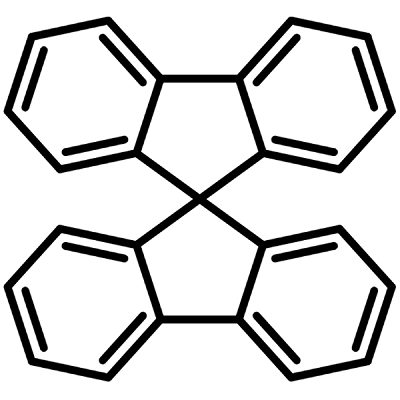
Structurally, 9,9′-Spirobifluorene (SBF), CAS number 159-66-0, can be considered as two fluorenes joined together at 9,9'-positions. 9,9'-Spirobifluorene has received considerable interest for organic electronics, featuring a sp3-hybridized carbon with two perpendicularly aligned π systems. The two fluorene units through a shared spiro-carbon are hence maintained in orthogonal planes.
Spiro compounds with 9,9′-spirobifluorene core of inherent rigid structure thus have attracted great attention as organic functional materials for their specific physical properties, such as high glass transition temperatures, good solubility and their amorphous nature. The prominent spiro-structure can also very effectively suppresses excimer formation frequently observed in the solid state of many fluorescent dyes. Particularly, spirobifluorenes are known for their excellent thermal and chemical stabilities, high quantum efficiencies, as well as nondispersive ambipolar carrier transporting properties.
9,9'-spirobifluorene-based conjugated microporous organic polymers (COPs) can be obtained directly from unsubstituted spirobifluorene using an inexpensive FeCl3 mediator via either oxidative polymerization, Friedel-Crafts polymerization, or competitive oxidative/Friedel-Crafts polymerization. Those COPs show large surface areas (940 – 1980 m2g-1) and high micropore volumes (0.5 – 0.9 cm3g-1).
General Information
| CAS Number | 159-66-0 |
| Chemical Formula | C25H16 |
| Full Name | 9,9′-Spirobifluorene |
| Molecular Weight | 316.39 g/mol |
| Synonyms | 9,9′-Spirobi[9H-fluorene], SBF |
| Classification / Family | Spirobifluorene, semiconductor synthesis intermediates, low band gap polymers, OLED, OFETs, organic photovoltaics |
Chemical Structure
Product Details
| Purity | >99% (HPLC) |
| Melting Point | Tm = 202 °C |
| Appearance | White powder/crystals |
MSDS Documentation
 9,9′-Spirobifluorene MSDS Sheet
9,9′-Spirobifluorene MSDS Sheet
Literature and Reviews
-
Hole Mobilities of 2,7- and 2,2′-Disubstituted 9,9′-Spirobifluorene-Based Triaryldiamines and Their Application as Hole Transport Materials in OLEDs, Y. Liao et al., Chem. Mater. , 19, 6350–6357 (2007); DOI: 10.1021/cm702230.
- 2-Substituted vs 4-substituted-9,90-spirobifluorene host materials for green and blue phosphorescent OLEDs: a structureeproperty relationship study, S. Thiery et al., Tetrahedorn, 70, 6337-6351 (2014); DOI: 10.1016/j.tet.2014.05.081.
- Synthesis, Structures, and Photoinduced Electron Transfer Reaction in the 9,9′-Spirobifluorene-Bridged Bipolar Systems, K. Wong et al., J. Org. Chem. 2006, 71, 456-465 (2006); DOI: 10.1021/jo0512047.
