IDTTB6-2Br
CAS Number 2227361-27-3
Chemistry Building Blocks, Dibromo Monomers, Heterocyclic Building Blocks, Materials, MonomersIDTTB6-2Br, used to synthesise the ITIC non-fullerene acceptors
High quality and expert support available online
Specifications | MSDS | Literature and Reviews
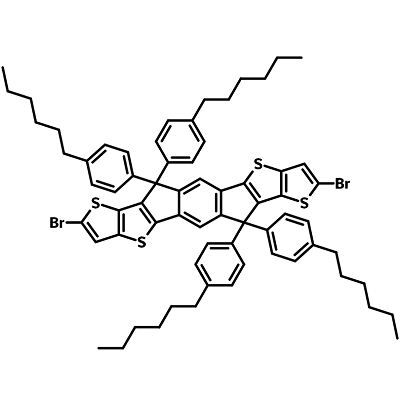
2,8-Dibromo-6,12-dihydro-6,6,12,12-tetrakis(4-hexylphenyl)-dithieno[2,3-d:2′,3′-d′]-s-indaceno[1,2-b:5,6-b′]dithiophene (IDTTB6-2Br), CAS number 2227361-27-3, is a useful, IDTT cored, intermediate for the synthesis of ITIC-series non-fullerene acceptors such as ITIC, ITIC-2F, ITIC-DCl, ITIC-M, ITIC-Th, and ITIC-DM. Such acceptors can be used to create highly efficient polymer organic solar cells.
IDTTB6-2Br has the structure of a highly conjugated fused indacenodithieno[3,2-b]thiophene (IDTT) core with four hexylphenyl attachments oriented almost perpendicularly to the backbone. The introduction of four hexylphenyl groups improves the solubility and degree of crystallinity in the film state.
We stock high purity (>98%) IDTTB6-2Br and a range of other non-fullerene acceptor monomers for priority dispatch (lead times may apply for large quantities).
Enabling facile coupling reaction
with bromide end groups
Improving solubility and crystallinity
With four hexylphenyl side chain
Worldwide shipping
Quick and reliable shipping
Electron-rich building block
Preparation of low bandgap non-fullerene acceptors (NFAs)
General Information
| CAS Number | 2227361-27-3 |
| Chemical Formula | C68H72Br2S4 |
| Molecular Weight | 1177.37 g/mol |
| Full Name | 2,8-Dibromo-6,12-dihydro-6,6,12,12-tetrakis(4-hexylphenyl)-dithieno[2,3-d:2′,3′-d′]-s-indaceno[1,2-b:5,6-b′]dithiophene |
| Synonyms | 2,8-Dibromo-6,6,12,12-tetrakis(4-hexylphenyl)-6,12-dihydro-dithieno[2,3-d:2',3'-d']-s-indaceno[1,2-b:5,6-b']dithiophene |
| Classification / Family | Indacenodithieno[3,2-b]thiophene (IDTT), Monomer and intermediates, None-fullerene acceptors (NFAs), NFA-OSCs, printing electronics |
Chemical Structure
Product Details
| Purity | >98% (by NMR) |
| Melting Point | N/A |
| Appearance | Yellow crystalline powder |
MSDS Documentation
Literature and Reviews
- Performance Optimization of Parallel-Like Ternary Organic Solar Cells through Simultaneous Improvement in Charge Generation and Transport, W. Hadmojo et al., Adv. Funct. Mater., 1808731 (2019); DOI: 10.1002/adfm.201808731.
- Donor polymer fluorination doubles the efficiency in non-fullerene organic photovoltaics, N. Bauer et al., . Mater. Chem. A, 5, 22536 (2017); DOI: 10.1039/c7ta07882j.
- Polymer Donors for High-performance Non-fullerene Organic Solar Cells, H. Fu et al., Angew. Chem. Int. Ed., 58 (14); 4442-4453 (2019); DOI: 10.1002/ange.201806291.

 IDTTB6-2Br MSDS Sheet
IDTTB6-2Br MSDS Sheet