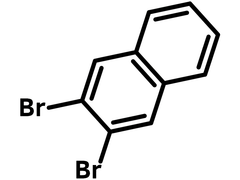
2,3-Dibromonaphthalene
CAS Number 13214-70-5
Chemistry Building Blocks, Materials, Monomers, Non-Heterocyclic Building BlocksDihalogenated naphthalene compound can be a suitable host for fluorescent polycyclic aromatic hydrocarbons molecules
Also used as a building block leading to more complex structures in application of organic electronics.
Specifications | MSDS | Literature and Reviews
2,3-Dibromonaphthalene (2,3-DBN), CAS number 13214-70-5, is another useful intermediate and isomer of dibromonaphthalene with two heavy bromo substituents sitting next to each other around the naphthalene ring.
X-ray diffraction analysis shows that the crystal structure of 2,3-dibromonaphthalene is ordered where the weak and directional forces generate the herringbone packing of the constituting molecules. For this reason, 2,3-dibromonaphthalene is employed as a suitable host for fluorescent polycyclic aromatic hydrocarbons (PAH) molecules. Single molecules of terrylene in 2,3-dibromonaphthalene crystallise also in a herringbone pattern, directed by the 2,3-dibromonaphthalene host.
Naphthalene building block
for the synthesis of polycyclic aromatic hydrocarbons and organic photovoltaic materials
Worldwide shipping
Quick and reliable shipping
Capped with bromides
for facile coupling reactions
High purity
>98% Purity
General Information
| CAS Number | 13214-70-5 |
| Chemical Formula | C10H6Br2 |
| Full Name | 2,3-Dibromonaphthalene |
| Molecular Weight | 285.97 g/mol |
| Synonyms | 2,3-DBN |
| Classification / Family | Naphthalenes, Semiconductor synthesis intermediates, Low band gap polymers, OLED, OFETs, organic photovoltaics |
Chemical Structure
Product Details
| Purity | >98% (1H NMR) |
| Melting Point | Tm = 142 °C |
| Appearance | White to off-white powder/crystals |
MSDS Documentation
 2,3-Dibromonaphthalene MSDS Sheet
2,3-Dibromonaphthalene MSDS Sheet
Literature and Reviews
- A Highly Warped Heptagon-Containing sp2 Carbon Scaffold via Vinylnaphthyl π-Extension, J. Farrell et al., Angew.Chem., 131, 16656–16659 (2019); DOI:10.1002/ange.201909975.
- Matrix-induced Linear Stark Effect of Single Dibenzoterrylene Molecules in 2,3-Dibromonaphthalene Crystal, A. Moradi et al., ChemPhysChem, 20,55–61 (2019); DOI: 10.1002/cphc.201800937.
- Spectra and nature of the electronic excited states of 2,3-dibromonaphthalene single crystals, I. Deperasińska et al., J. Lumin., 213, 108-116 (2019); DOI: 10.1016/j.jlumin.2019.05.011.
