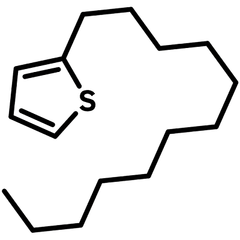
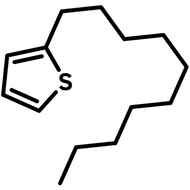
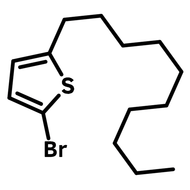
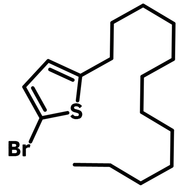
2-Dodecylthiophene
CAS Number 4861-61-4
Chemistry Building Blocks, Heterocyclic Building Blocks, Monomers
An intermediate leading to more complex structures
With the tendency to aggregate and self-aggregate, it gives rise to orderly packed molecules in film.
2-Decylthiophene (CAS number 4861-61-4) with a dodecyl (C12H25) carbon hydrate side chain, is an useful alkylated thiophene that is commonly used as an intermediate for the construction of semiconducting polymers or small molecules for device fabrication of organic field-effect transistors and polymer solar cells.
PTVTTT2T, a copolymer with a backbone of biaxially 2-dodecylthiophene side chained thieno[3,2-b]thiophene (TT2T) and thienylene-vinylene-thienylene (TVT), possessed better molecular packing in thin films with a nanofiber structure owing to its coplanar backbone and the optimum side chain length for solubility and film morphology. The average field-effect charge mobility of PTVTTT2T was found to be 0.64 cm2 V-1 s-1 with a high on/off ratio over 107 under ambient environment for over three months.
General Information
| CAS Number | 4861-61-4 |
| Chemical Formula | C16H28S |
| Full Name | 2-Decylthiophene |
| Molecular Weight | 252.46 g/mol |
| Synonyms | N/A |
| Classification / Family | Thiophene, Semiconductor synthesis intermediates, Low band gap polymers, OLED, OFETs, organic photovoltaics |
Chemical Structure

Product Details
| Purity | >98% (1H NMR) |
| Boiling Point | Tm = 155 °C at 2 mmHg |
| Appearance | Light yellow to orange liquid |
| Relative Density | 0.91 |
MSDS Documentation
Literature and Reviews
- Biaxially Extended Conjugated Polymers with Thieno[3,2-b]thiophene Building Block for High Performance Field-Effect Transistor Applications, P. Chao et al., Macromolecules, 48, 16, 5596–5604 (2015); DOI: 10.1021/acs.macromol.5b01243.
- Star-like substituted hexaarylbenzenes: synthesis and mesomorphic properties, Y. Geng et al., J. Mater. Chem., 11, 1634-1641 (2011); DOI: 10.1039/B101163O.
- Effect of lipid on formation of Maillard and lipid-Maillard meaty flavour compounds in heated cysteine-xylose-methyl linoleate system, Y. Wang et al., Flavour Fragr. J., 37 (5), 274-284 (2022); DOI: 10.1002/ffj.3710.
- New Synthesis of 3,6-Dibromophthalonitrile and Phthalocyanine Having Eight Thienyl Substituents at Peripheral α-Positions, R. Wang et al., J. Hetero. Chem., 52 (4), 1230-1233 (2015); DOI: 10.1002/jhet.2130.
- Benzodithiophene homopolymers synthesized by Grignard metathesis (GRIM) and Stille coupling polymerizations, H. Magurudeniya et al., J. Mater. Chem. A, 2, 8773-8781 (2014); DOI: 10.1039/C4TA01739K.
- Synthesis, characterization and photovoltaics studies of 3-alkylthienyl thiophene based polymers prepared via direct arylation polymerization, G. Saini et al., 8 (7), 813-818 (2017); Adv. Mater. Lett., DOI: 10.5185/amlett.2017.1483.
- Trans–cis isomerization assisted synthesis of solution-processable yellow fluorescent maleic anhydrides for white-light generation, M. Ozdemir et al., Synth. Met., 210 (B), 192-200 (2015); DOI: 10.1016/j.synthmet.2015.09.027.
- Design, synthesis, and characterization of α,ω-disubstituted indeno[1,2-b]fluorene-6,12-dione-thiophene molecular semiconductors. Enhancement of ambipolar charge transport through synthetic tailoring of alkyl substituents, M. Ozdemir et al., RSC Adv., 6, 212-226 (2016); DOI: 10.1039/C5RA22359H.
- Highly Contorted 1,2,5-Thiadiazole-Fused Aromatics for Solution-Processed Field-Effect Transistors: Synthesis and Properties, X. Shi et al., Chem. Asian J., 11 (15), 2188-2200 (2016); DOI: 10.1002/asia.201600675.
- Alkylated Fullerene Derivatives for Solution-Processable Organic Thin-Film Transistors and Bulkheterojunction Solar Cells, J. Nanosci. Nanotechnol., 14 (3), 2515-2519(2014); DOI: 10.1166/jnn.2014.8501.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
 2-Dodecylthiophene MSDS Sheet
2-Dodecylthiophene MSDS Sheet
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
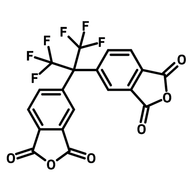
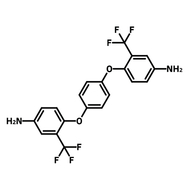
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
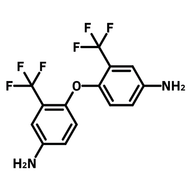
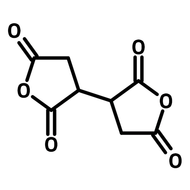
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)