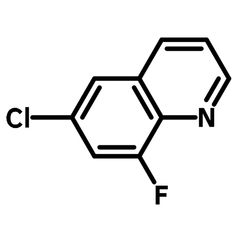
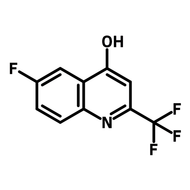
6-Chloro-8-fluoroquinoline
CAS Number 52200-53-0
Chemistry Building Blocks, Fluorinated Building Blocks, Heterocyclic Building Blocks, Monomers
A fluorinated heterocyclic building block
Used as a synthesis intermediate for APIs, DSSCs and sensors
6-Chloro-8-fluoroquinoline (CAS number 52200-53-0) is a fluorinated quinoline derivative with a chlorine and a fluorine at 6- and 8-position respectively. This building block is best known for its used in active pharmaceutical ingredients (APIs), such as Besifloxacin and Clinafloxacin in topical antibiotic treatments and conjunctive bacterial infections. Other than pharmaceutical uses, 6-Chloro-8-fluoroquinoline is a synthesis intermediate for Schiff bases, through nucleophilic aromatic substitution. These Schiff bases are used for metal-ion recognition sensors because their fluorescent emissions shift after chelating to a metal centre.

The Schiff bases formed by this building block are typically bidentate/tridentate ligands which readily coordinate to Ru and Ir ion centres to form dye complexes in applications of dye-sensitized solar cells(DSSCs).
Multiple functional groups
For facile synthesis
Worldwide shipping
Quick and reliable shipping
Fluorinated quinoline building block
For drug discoveries, dyes, and solar cells
High purity
>98% High purity
General Information
| CAS Number | 52200-53-0 |
| Chemical Formula | C9H5ClFN |
| Full Name | 6-Chloro-8-fluoroquinoline |
| Molecular Weight | 181.59 g/mol |
| Synonyms | N/A |
| Classification / Family | Fluorinated building block, Heterocyclic building block, Quinoline derivatives, Schiff base, Dyes, Semiconductor synthesis intermediates, organic photovoltaics |
Chemical Structure

Product Details
| Purity | >98% |
| Melting Point | Tm = 79 °C – 81 °C |
| Appearance | White to yellow crystal |
MSDS Documentation
 6-Chloro-8-fluoroquinoline MSDS Sheet
6-Chloro-8-fluoroquinoline MSDS Sheet
Literature and Reviews
-
Besifloxacin ophthalmic suspension 0.6% in the treatment of bacterial conjunctivitis patients with Pseudomonas aeruginosa infections, B. Silverstein et al., Clin. Ophthalmol., 6, 1987-1996(2012); DOI: 10.2147/OPTH.S35715.
-
Clinafloxacin for the treatment of bacterial endocarditis, D. Levine et al., Clin. Infect. Dis., 38(5), 620-631(2004); DOI: 10.1086/381670.
-
Copper(II) complexes based on quinoline-derived Schiff-base ligands: synthesis, characterization, HSA/DNA binding ability, and anticancer activity, Med. Chem. Commun., 9, 1663–1672(2018); DOI: 10.1039/c8md00223a.
-
A fluorescent sensor based on binaphthol-quinoline Schiff base for relay recognition of Zn2+ and oxalate in aqueous media, L. Tang et al., J. Chem. Sci., 128(8), 1337-1343(2016); DOI: 10.1007/s12039-016-1124-y.
-
Enhanced in vivo wound healing efficacy of a novel hydrogel loaded with copper(II) Schiff base quinoline complex(CuSQ) solid lipid nanoparticles, D. Abou El-ezz et al., Pharmaceuticals, 15(8), 978(2022); DOI: 10.3390/ph15080978.
-
Nano-synthesis, characterization and spectroscopic studies of chromium (III) complex derived from new quinoline-2-one for solar cell fabrication, A. Salman., J. Phys.: Conf. Ser., 1853, 012021(2021); DOI: 10.1088/1742-6596/1853/1/012021/
-
Ruthenium-triphenylphosphine complex with pendent quinolyl Schiff base ligand: synthesis, spectral characterization and catalytic property, S. Kundu et al., J. Indian Chem. Soc., 92, 1847-1853(2015); DOI: 10.5281/zenodo.5599621.
- Theoretical and experimental investigations of large stokes shift fluorophores based on a quinoline scaffold, B. Czaplińska et al., Molecules, 25, 2488(2020); DOI: 10.3390/molecules25112488.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
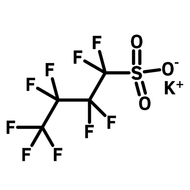
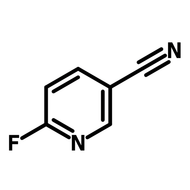
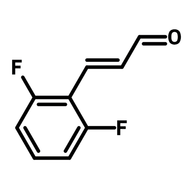
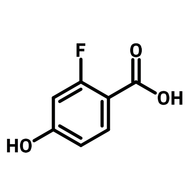
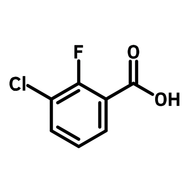
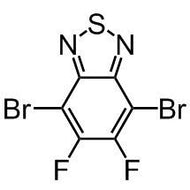
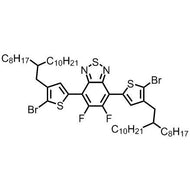
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)