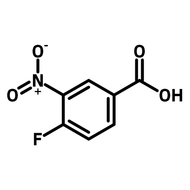
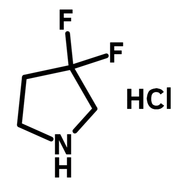
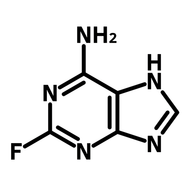
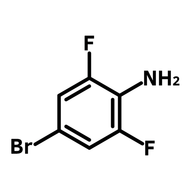
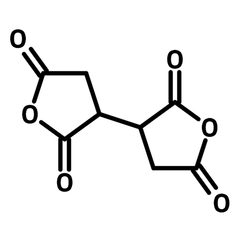
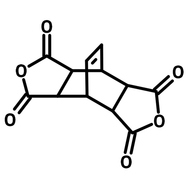
1,2,3,4-Butanetetracarboxylic dianhydride (BDA)
CAS Number 4534-73-0
Chemistry Building Blocks, COF Ligands, Diamines and Dianhydrides, Heterocyclic Building Blocks, Monomers, Porous Organic Frameworks
A dianhydride building block
An intermediate for polyester and polyimides synthesis in application of super absorbances, water treatments and wrinkle-free fabrics
1,2,3,4-Butanetetracarboxylic dianhydride (BDA, CAS number 4534-73-0) has two succinic anhydrides bridged by a carbon-carbon single bond. 1,2,3,4-Butanetetracarboxylic dianhydride readily reacts with amines and hydroxyls forming imides and esters. BDA is a well-known anti-wrinkle reagent to treat cotton fabrics, as its ability to crosslink the cellulose (through esterification). The breaking and tearing strengths of the fabrics are improved via crosslinking modification. Super absorbance hydrogels can be synthesized from polysaccharide and 1,2,3,4-butanetetracarboxylic dianhydride. The products are applied in water purification and agricultural water retention.
High ionic conductive ionomer can be prepared from BDA and diol. The ionomer is promising to use as solid polymer electrolytes.
General Information
| CAS Number | 4534-73-0 |
| Chemical Formula | C8H6O6 |
| Full Name | 1,2,3,4-Butanetetracarboxylic dianhydride |
| Molecular Weight | 198.13 g/mol |
| Synonyms | Tetrahydro(3,3'-bifuran)-2,2',5,5'-tetrone, 3-(2,5-Dioxooxolan-3-yl)oxolane-2,5-dione |
| Classification / Family | Dianhydride building block, Polyesters, Polyimides, Hydrogels, Absorbances |
Chemical Structure

Product Details
| Purity | >99% |
| Melting Point | Tm = 242 °C – 243 °C |
| Appearance | White to pale yellow to slight brown powder/crystal |
MSDS Documentation
 1,2,3,4-butanetetracarboxylic dianhydride (BDA) MSDS Sheet
1,2,3,4-butanetetracarboxylic dianhydride (BDA) MSDS Sheet
Literature and Reviews
-
Adsorption of Pb(II) ions from contaminated water by 1, 2, 3, 4-butanetetracarboxylic acid-modified microcrystalline cellulose: isotherms, kinetics, and thermodynamic studies, A. Hashem et al., Int. J. Biol. Macromol., 164, 3193-3203(2020); DOI: 10.1016/j.ijbiomac.2020.08.159.
-
Bio-synthesis of itaconic acid as an anti-crease finish for cellulosic fiber fabric, A. Boondaeng et al., RSC Adv., 11, 25943(2021); DOI: 10.1039/d1ra05037k.
-
Green routes for the development of chitin/chitosan sustainable hydrogels, M. Maddaloni et al., Sus. Chem., 1, 325–344(2020); DOI: 10.3390/suschem1030022.
-
Ionomers from step-growth polymerization: highly ordered ionic aggregates and ion conduction, L. Yan et al., Macromolecules, 53, 1777−1784(2020); DOI: 10.1021/acs.macromol.9b02220.
-
Polysaccharide based superabsorbent hydrogels and their methods of synthesis: A review, M. Qureshi et al., Carbohydrate Polymer Technologies and Applications, 1, 100014(2020); DOI: 10.1016/j.carpta.2020.100014.
-
Role of crosslinkers for synthesizing biocompatible, biodegradable and mechanically strong hydrogels with desired release profile, S. Zafar et al., Polym. Bull., 79, 9199–9219 (2022); DOI: 10.1007/s00289-021-03956-8.
- Superabsorbent polymers used for agricultural water retention, L. Chang et al., Polym. Test., 94, 107021(2021); DOI: 10.1016/j.polymertesting.2020.107021.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)