2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine
CAS Number 84-67-3
Chemistry Building Blocks, COF Ligands, Diamines and Dianhydrides, Monomers, Non-Heterocyclic Building Blocks, Porous Organic Frameworks
A rigid biphenyl diamine building block
For the synthesis of polyimides and enamines in applications of solar cells, telecommunication applications, batteries, and separation membranes
2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine, CAS number 84-67-3, is a biphenyl derivative with two amines and two methyl groups. 2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine reacts with diphenylacetaldehyde catalysed by camphorsulfonic acid forming an enamine through nucleophilic substitution and dehydration reaction. The fully conjugated enamine is a p-type semiconductor used for hole transport layers (hole mobility 2.5 × 10-2 cm2/Vs) in solar cells (efficiency 18.4%).
2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine is also used to synthesize low dielectric constant and low dielectric loss polyimides for telecommunication application with fast transport speed and low signal loss. The low dielectric property is achieved by the branched methyl groups enlarging the intermolecular spacing, hence the effective number of dipole moments is reduced.
General Information
| CAS Number | 84-67-3 |
| Chemical Formula | C14H16N2 |
| Full Name | 2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine |
| Molecular Weight | 212.29 g/mol |
| Synonyms | m-Tolidine, 2,2'-Dimethylbenzidine, 4,4'-Diamino-2,2'-dimethylbiphenyl, 4-(4-Amino-2-methylphenyl)-3-methylbenzenamine |
| Classification / Family | Diamine building block, Solar cells, Hole transport, Polyimides, Sensors, Telecommunication |
Chemical Structure
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine chemical structure, CAS 84-67-3](https://cdn.shopify.com/s/files/1/0823/0287/files/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-body_240x180.png?v=1680597263)
Product Details
| Purity | >99% |
| Melting Point | Tm = 106 °C |
| Appearance | White to off-white powder |
MSDS Documentation
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](https://cdn.shopify.com/s/files/1/0823/0287/files/msds-sheets_60x60.jpg) 2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine MSDS Sheet
2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine MSDS Sheet
Literature and Reviews
-
Enamine-based hole transporting materials for vacuum-deposited perovskite solar cell, M. Steponaitis et al., Sustainable Energy Fuels, 4, 5017-5023(2020); DOI: 10.1039/D0SE00728E.
-
Intrinsic low-dielectric constant and low-dielectric loss aliphatic-aromatic copolyimides: The effect of chemical structure, J. Lee et al., Mater. Today Commun., 33, 104479(2022); DOI: 10.1016/j.mtcomm.2022.104479.
-
Orientation control of the microphase-separated nanostructures of block copolymers on polyimides substrates, H. Maeda et al., J. Photopolym. Sci. Technol., 34(5), 439-448(2021); DOI: 10.2494/photopolymer.34.439.
-
Polyimide-coated glass microfiber as polysulfide per-selective separator for high-performance lithium-sulphur batteries, M.-J. Kim et al., Nanomatetrials, 9, 1612(2019); DOI: 10.3390/nano9111612.
-
Recent advances of thin film composite membranes for pervaporation applications: A comprehensive review, T. Zhu et al., Adv. Membr., 1, 100008(2021); DOI: 10.1016/j.advmem.2021.100008.
- Solution-processable colorless polyimides derived from hydrogenated pyromellitic dianhydride: strategies to reduce the coefficients of thermal expansion by maximizing the spontaneous chain orientation behavior during solution casting, M. Hasegawa et al., Polymers, 14, 1131(2022); DOI: 10.3390/polym14061131.
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine CAS 84-67-3](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=240)
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
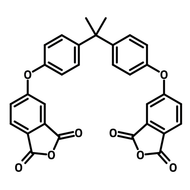
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
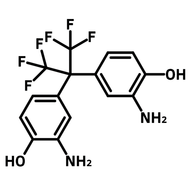
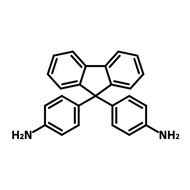
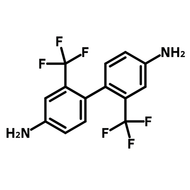
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)