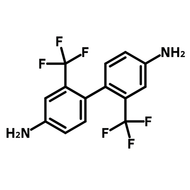
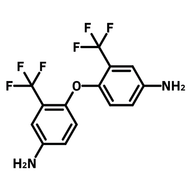
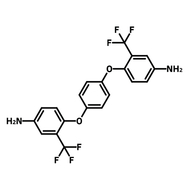
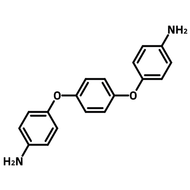
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde
CAS Number 150443-85-9
Carbaldehyde Monomers, COF Ligands, Materials, Monomers, Porous Organic Frameworks
Covalent Organic Frameworks (COFs) Biphenyl Ligand
A linear linker for COFs in applications of effective chromium adsorption and carbon dioxide reduction
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde (TABP), CAS number 150443-85-9, is consisted of a biphenyl linker with four carbaldehyde groups at 3,3',5,5'-positions of the biphenyl structure.
Derived from [1,1'-biphenyl]-3,3',5,5'-tetracarbaldehyde and bearing hydroxyl groups, dual-pore COF-BTA-DHBZ showed a fast removing process of chromium (VI) with a maximum adsorption capacity up to 384 mg g−1. Study revealed that the hydroxyl groups on the backbone of the COF played a critical role for the capture of Cr (VI).
A donor-acceptor (D–A) structure of Co-COF-BT, comprising (1,1′-biphenyl)-3,3′,5,5′-tetracarbaldehyde and 4,4′-(2,1,3-benzothiadiazole-4,7-diyl)bis[benzenamine], possesses excellent photocatalytic CO2 reduction activity with a CO yield of 2423 μmol g−1h−1 and selectivity >99% under visible light. It is believed that the introduction of Co2+ to the COF pores provides additional active sites. It is the Co-S bond in Co-COF-BT that generates an additional channel for ultrafast electron migration from the COF-BT unit to the Co2+ active centre to increase charge mobility and reduce the energy barrier.
MOF and COF ligands
Aldehyde ligand for cross-linked COF networks
Worldwide shipping
Quick and reliable shipping
High purity
>98% High purity
Facile reactions
Aldehyde possesses excellent reactivity
General Information
| CAS Number | 150443-85-9 |
|---|---|
| Chemical Formula | C16H10O4 |
| Full Name | [1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde |
| Molecular Weight | 266.25 g/mol |
| Synonyms | TABP, BTA, 1,1'-Biphenyl-3,3',5,5'-tetracarbaldehyde |
| Classification / Family | Biphenyls, COF ligands |
Chemical Structure

Product Details
| Purity | >98% |
|---|---|
| Melting Point | N/A |
| Appearance | White to light yellow powder/crystals |
MSDS Documentation
 [1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde MSDS Sheet
[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde MSDS Sheet
Literature and Reviews
-
Cheap transition metal reinforced donor–acceptor covalent organic frameworks for CO2 photoreduction, L. Ai et al, Chem. Eng. J., 475, 146106 (2023); DOI: /10.1016/j.cej.2023.146106.
-
Efficient Removal of Cr(VI) from Aqueous Solutions by a Dual-Pore Covalent Organic Framework, F. Cui et al., Adv. Sustainable Syst., 3 (4), 1800150 (2019); DOI: 10.1002/adsu.201800150.
-
2D Covalent Organic Frameworks with Kagome Lattice: Synthesis and Applications, J. Tu et al., Chem, Eur. J., e202302380 (2023); DOI: 10.1002/chem.202302380.
-
Pd-Immobilized Schiff Base Double-Layer Macrocycle: Synthesis, Structures, Peroxidase Mimic Activity, and Antibacterial Performance, K. Wang et al., ACS Appl. Mater. Interfaces, 14 (1), 1423–1433 (2022); DOI: 10.1021/acsami.1c19795.
-
Covalent Organic Frameworks: Structures, Synthesis, and Applications, M. Lohse et al., Adv. Funct. Mater., 28, 1705553 (2018); DOI: 10.1002/adfm.201705553.
-
Covalent Organic Frameworks: Design, Synthesis, and Functions, K. Geng et al., Chem. Rev., 120 (16), 8814–8933 (2020); DOI: 10.1021/acs.chemrev.9b00550.
- Two-dimensional dual-pore covalent organic frameworks obtained from the combination of two D2h symmetrical building blocks, Y. Tian et al., Chem. Commun., 52, 11704-11707 (2016); DOI: 10.1039/C6CC06637B.
Related Products
We stock a wide range of COF ligands available to purchase online. Please contact us if you cannot find what you are looking for.
![[1,1'-Biphenyl]-3,3',5,5'-tetracarbaldehyde CAS 150443-85-9](http://www.ossila.com/cdn/shop/files/bta-150443-85-9-chemical-structure-title.jpg?v=1700739580&width=240)
![4',4''',4'''''-(1,3,5-triazine-2,4,6-triyl)tris([1,1'-biphenyl]-4-amine)](http://www.ossila.com/cdn/shop/files/tabpt-2130745-76-3-chemical-structure-title.jpg?v=1708616414&width=190)
![1,3,5-Tris[4-amino(1,1-biphenyl-4-yl)]benzene](http://www.ossila.com/cdn/shop/files/tabpb-1400987-00-9-chemical-structure-title.jpg?v=1708610226&width=190)
![4',4''',4'''''-(1,3,5-Triazine-2,4,6-triyl)tris([1,1'-biphenyl]-4-carbaldehyde)](http://www.ossila.com/cdn/shop/files/tfbptz-1221509-80-3-chemical-structure-title.jpg?v=1708093553&width=190)
![[1,1':4',1'':4'',1'''-Quaterphenyl]-4,4'''-diamine](http://www.ossila.com/cdn/shop/files/qpda-53693-67-7-chemical-structure-title.jpg?v=1708086116&width=190)
![5''-(4'-Formyl-[1,1'-biphenyl]-4-yl)-[1,1':4',1'':3'',1''':4''',1''''-quinquephenyl]-4,4''''-dicarbaldehyde](http://www.ossila.com/cdn/shop/files/tfbpb-805246-78-0-chemical-structure-title.jpg?v=1708014325&width=190)
![4',4''',4''''',4'''''''-(Ethene-1,1,2,2-tetrayl)tetrakis([1,1'-biphenyl]-4-carbaldehyde)](http://www.ossila.com/cdn/shop/files/tapbe-1624970-54-2-chemical-structure-title.jpg?v=1707929446&width=190)
![[1,1':3',1''-Terphenyl]-3,3'',5,5''-tetracarbaldehyde](http://www.ossila.com/cdn/shop/files/tpta-201734-76-1-chemical-structure-title.jpg?v=1707927322&width=190)
![4',5'-Bis(4-aminophenyl)-[1,1':2',1''-terphenyl]-4,4''-diamine](http://www.ossila.com/cdn/shop/files/baptpda-2458125-05-6-chemical-structure-title.jpg?v=1707921329&width=190)
![Tris[4-(5-formyl-2-thienyl)phenyl]amine](http://www.ossila.com/cdn/shop/files/tta-tpa-883236-47-3-chemical-structure-title.jpg?v=1707918937&width=190)
![2,9-Bis[p-(formyl)phenyl]-1,10-phenanthroline](http://www.ossila.com/cdn/shop/files/bfpp-120085-99-6-chemical-structure-title.jpg?v=1707839745&width=190)
![[1,1':4',1''-Terphenyl]-4,4''-diacetonitrile](http://www.ossila.com/cdn/shop/files/tpdan-1000559-50-1-chemical-structure-title.jpg?v=1702310114&width=190)
![2,4,6-Tris[4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]-1,3,5-triazine](http://www.ossila.com/cdn/shop/files/tbptrz-1447947-87-6-chemical-structure-title.jpg?v=1702305933&width=190)
![2',5'-Dimethoxy-[1,1':4',1''-terphenyl]-4,4''-dicarbaldehyde](http://www.ossila.com/cdn/shop/files/tpda-ome-111759-27-4-chemical-structure-title.jpg?v=1702297940&width=190)
![1,3,5-Tris[(4-phenylboronic acid)]benzene](http://www.ossila.com/cdn/shop/files/tpbab-900795-73-5-chemical-structure-title.jpg?v=1702293555&width=190)
![5'-(4-Formylphenyl)-2',4',6'-trimethyl-[1,1':3',1''-terphenyl]-4,4''-dicarbaldehyde](http://www.ossila.com/cdn/shop/files/tmtfpb-2195343-70-3-chemical-structure-title.jpg?v=1702046856&width=190)
![4,4'-(Benzo[c][1,2,5]thiadiazole-4,7-diyl)dianiline](http://www.ossila.com/cdn/shop/files/btda-1203707-77-0-chemical-structure-title.jpg?v=1702045373&width=190)
![4,4'-(Benzo[c][1,2,5]thiadiazole-4,7-diyl)dibenzaldehyde](http://www.ossila.com/cdn/shop/files/btdba-914651-17-5-chemical-structure-title.jpg?v=1702044113&width=190)
![4',4''',4'''''-Nitrilotris([1,1'-biphenyl]-4-carbaldehyde)](http://www.ossila.com/cdn/shop/files/ntbca-872689-79-7-chemical-structure-title.jpg?v=1701944784&width=190)
![5'-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-[1,1':3',1''-terphenyl]-4,4''-diamine](http://www.ossila.com/cdn/shop/files/5-4-4-5-5-tetramethyl-1-3-2-dioxaborolan-2-yl-1-1-3-1-terphenyl-4-4-diamine-chemical-structure-title.png?v=1700149769&width=190)
![5'-(4,4,5,5-Tetramethyl-1,3,2-dioxaborolan-2-yl)-[1,1':3',1''-terphenyl]-4,4''-dicarbaldehyde](http://www.ossila.com/cdn/shop/files/5-4-4-5-5-tetramethyl-1-3-2-dioxaborolan-2-yl-1-1-3-1-terphenyl-4-4-dicarbaldehyde-chemical-structure-title.png?v=1700133486&width=190)
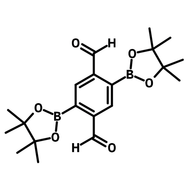
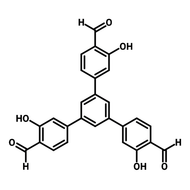
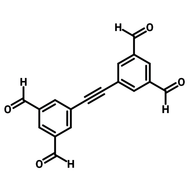
![3,3'-Dihydroxy-[1,1'-biphenyl]-4,4'-dicarbaldehyde](http://www.ossila.com/cdn/shop/files/3-3-dihydroxy-1-1-biphenyl-4-4-dicarbaldehyde-chemical-structure-title.png?v=1699879460&width=190)
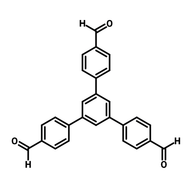
![2,2'-(5'-(4-(Cyanomethyl)phenyl)-[1,1':3',1''-terphenyl]-4,4''-diyl)diacetonitrile](http://www.ossila.com/cdn/shop/files/2-2-5-4-cyanomethylphenyl-1-1-3-1-terphenyl-4-4-diyldiacetonitrile-chemical-structure-title.png?v=1700225015&width=190)
![[3,3-Bipyridine]-6,6-dicarboxaldehyde](http://www.ossila.com/cdn/shop/files/3-3-bipyridine-6-6-dicarboxaldehyde-chemical-structure-title.png?v=1698223775&width=190)
![[1,1'-Biphenyl]-3,4',5-tricarbaldehyde](http://www.ossila.com/cdn/shop/files/1-1-biphenyl-3-4-5-tricarbaldehyde-chemical-structure-title.png?v=1697627856&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)