PBDTTTPD, donor material used in the application of polymer solar cells
High purity polymer available for fast, secure dispatch
PBDTTTPD, poly[(5,6-dihydro-5-octyl-4,6-dioxo-4H-thieno[3,4-c]pyrrole-1,3-diyl)[4,8-bis[5-(2-ethylhexyl)-2-thienyl]benzo[1,2-b:4,5-b']dithiophene-2,6-diyl], CAS number 1426534-44-2, has been used in the application of all polymer solar cells with PBDTTTPD as polymer donor and PNDI(2HD)T polymer acceptor providing high power-conversion efficiency of 6.64% [1].
PBDTTTPD shows complementary absorption with poly-naphthalene-diimides as acceptor polymers and the low-lying HOMO energy level of PBDTTTPD produces a high VOC value in all-PSCs. Polymer-polymer solar cell with PBDTT-TPD as donor and PNF222 (PNDI(2OD)2T-2F) as acceptor has a VOC = 1.00 V, JSC = 11.68 mA cm−2 and FF = 0.52 to give a PCE 0f 6.09% [2].
Luminosyn™ PBDTTTPD
Luminosyn™ PBDTTTPD is now available.
High purity
PBDTTTPD is purified by soxhlet extraction with methanol, hexane and chloroform under argon atmosphere
Batch-specific GPC data
Have confidence in what you are ordering; batch-specific GPC data for your thesis or publications
Large quantity orders
Plan your experiments with confidence with polymers from the same batch
General Information
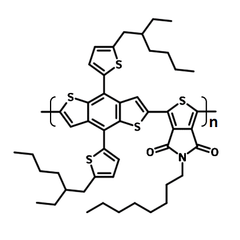
| Full name | Poly[(5,6-dihydro-5-octyl-4,6-dioxo-4H-thieno[3,4-c]pyrrole-1,3-diyl)[4,8-bis[5-(2-ethylhexyl)-2-thienyl]benzo[1,2-b:4,5-b']dithiophene-2,6-diyl ]] |
| Synonyms | PBDTT(EH)-TPD(Oct), PBDTT-TPD |
| CAS number | 1426534-44-2 |
| Chemical formula | (C48H57NO2S5)n |
| Molecular weight | See Batch Details table above |
| HOMO / LUMO | HOMO = - 5.49 eV, LUMO = - 3.47 eV [1] |
| Solubility | Chloroform, chlorobenzene, dichlorobenzene |
| Classification / Family | Benzodithiophene, BDT, Thienopyrrole, Heterocyclic five-membered ring, Organic semiconducting materials, Low band gap polymers, Organic photovoltaics, All-polymer solar cells, OFETs, Photodetectors |
Chemical Structure

MSDS Documentation
Pricing
| Batch | Quantity | Price |
| M2054A | 100 mg | £280 |
| M2054A | 250 mg | £580 |
| M2054A | 500 mg | £1050 |
| M2054A | 1 g | £1750 |
| M2054A | 5 g / 10 g* | Please enquire |
*for 5 - 10 grams order quantity, the lead time is 4-6 weeks
Batch Details
| Batch | Mw | Mn | PDI | Stock Info |
| M2054A1 | 37,952 | 13,848 | 2.74 | Low Stock |
| M2054A2 | 27,925 | 9,673 | 2.89 | In Stock |
Literature and Reviews
- Flexible, highly efficient all-polymer solar cells, T. Kim et al., Nat. Commun. 6, 8547 (2015); doi:10.1038/ncomms9547.
- Controlling Energy Levels and Blend Morphology for All-Polymer Solar Cells via Fluorination of a Naphthalene Diimide-Based Copolymer Acceptor, M. A. Uddin et al., Macromolecules 2016, 49, 6374−6383 (2016); DOI: 10.1021/acs.macromol.6b01414.
- Toward Highly Sensitive Polymer Photodetectors by Molecular Engineering, L. Zhang et al., Adv. Mater., 27, 6496–6503 (2015); DOI: 10.1002/adma.201502267.n2200.
 PBDTTTPD MSDS Sheet
PBDTTTPD MSDS Sheet