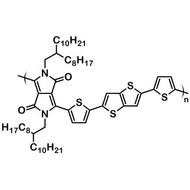
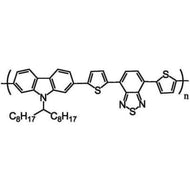
F8T2
CAS Number 210347-56-1
Chemistry Building Blocks, Interface Polymers, Luminosyn™ Polymers, OFET & OLED Polymer Materials, OPV Polymers, Semiconducting Polymers
F8T2, popular semiconducting polymer used in organic electronics
High purity and available online for priority dispatch
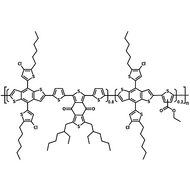
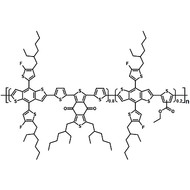
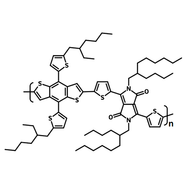
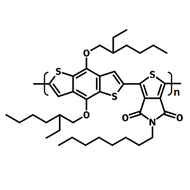
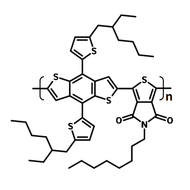
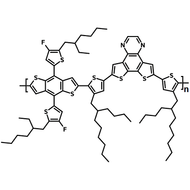
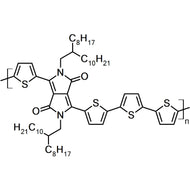
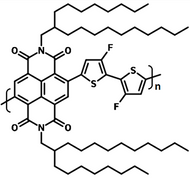
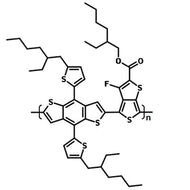
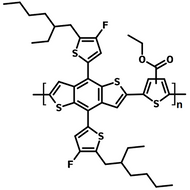
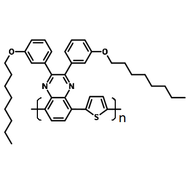
Poly(9,9-dioctylfluorene-alt-bithiophene), also known as F8T2 (CAS number 210347-56-1), is a semiconducting material that is widely used in organic electronics such as organic photovoltaics, polymer light-emitting diodes (PLED) and organic field-effect transistors (OFETs). Comparing with poly-3-hexylthiophene, F8T2 has even higher mobilities of 0.1 cm2/V·s and relatively higher stability against chemical doping by environmental oxygen or residual impurities such as mobile sulphonic acid in the PEDOT:PSS ink. This enables devices with higher on-off current ratios exceeding 105 and with better operating stability than printed poly-3-hexylthiophene devices[1].
The absorption in the blue region of F8T2 makes it an excellent donor polymer to blend with an acceptor having complementary spectrum or assemble a tandem cell with other low bandgap-conjugated polymers with absorption extended in the red region.
Luminosyn™ F8T2
Luminosyn™ F8T2 is now available.
High molecular weight and high purity
F8T2 is purified via Soxhlet extraction with methanol, hexane and chloroform under an argon atmosphere
Good solubility
Good solubility in most of common solvents (toluene, chloroform and chlorobenzene)
Large quantity orders
Plan your experiments with confidence with polymers from the same batch
General Information
| CAS Number | 210347-56-1 |
|---|---|
| Chemical Formula | (C37H44S2)n |
| Molecular Weight | See batch information for details |
| HOMO / LUMO | HOMO = 5.5 eV / LUMO = 3.1 eV [1] |
| Synonyms | PFOT, Poly(9,9-dioctylfluorene-alt-bithiophene), Poly[(9,9-dioctylfluorenyl-2,7-diyl)-co-bithiophene] |
| Classification / Family | Polyfluorenes, Bithiophenes, Heterocyclic five-membered ring, Organic semiconducting materials, PLED green emitter materials, Organic Photovoltaics, Polymer Solar Cells, Light-emitting Diodes, OFET materials |
| Suggested Solvents | Chloroform, chlorobenzene or dichlorobenzene |
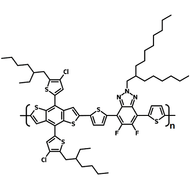
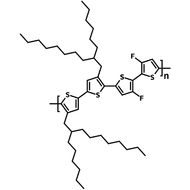
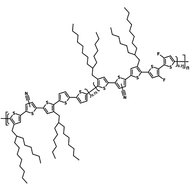
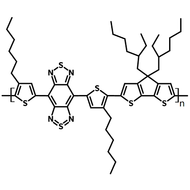
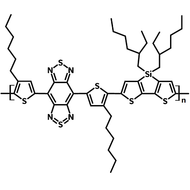
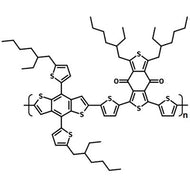
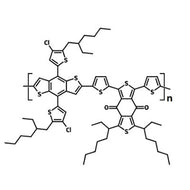
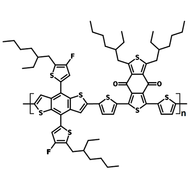
Chemical Structure

Device Structure(s)
| Device structure | ITO/PEDOT:PSS/TFB/F8T2/Ca [3] |
|---|---|
| Colour | Green |
| Max. Luminance | 23,400 |
| Max. Current Efficiency | 3.68 cd/A |
| Max. Power Efficiency | 2.9 lm W−1 |
MSDS Documentation
Pricing
| Batch | Quantity | Price |
|---|---|---|
| M0501A | 250 mg | £310 |
| M0501A | 500 mg | £560 |
| M0501A | 1 g | £1000 |
| M0501A | 5 g / 10 g* | Please enquire |
*For 5 - 10 grams order quantity, the lead time is 4-6 weeks.
Batch information
| Batch | Mw | Mn | PDI | Stock info |
| M502 | 136,320 | 53,866 | 2.53 | Discontinued |
| M503 | 45,586 | 16,493 | 2.76 | Discontinued |
| M0501A1 | 63,525 | 26,387 | 2.41 | Discontinued |
| M0501A2 | 116,093 | 45,136 | 2.6 | In Stock |
Literature and Reviews
- Annealing effect of polymer bulk heterojunction solar cells based on polyfluorene and fullerene blend, J-H. Huang et al., Org. Electronics, 10, 27–33 (2009), doi:10.1016/j.orgel.2008.09.007.
- High-Efficiency Polymer LEDs with Fast Response Times Fabricated via Selection of Electron-Injecting Conjugated Polyelectrolyte Backbone Structure, M. Suh et al., ACS Appl. Mater. Interfaces, (2015), DOI: 10.1021/acsami.5b07862.
- On the use and influence of electron-blocking interlayers in polymer light-emitting diodes, R. Jin et al., Phys. Chem. Chem. Phys., 11, 3455-3462 (2009). DOI: 10.1039/B819200F.
- High-Resolution Inkjet Printing of All-Polymer Transistor Circuits, H. Sirringhaus et al., Science, 290 (5499), 2123-2126 (2000), DOI: 10.1126/science.290.5499.2123.
- Organic Light-Emitting Diodes Based on Poly(9,9-dioctylfluorene-co-bithiophene) (F8T2), P. Levermore et al., Adv. Funct. Mater., 19, 950–957 (2009); DOI: 10.1002/adfm.200801260.
- Mobility enhancement in conjugated polymer field-effect transistors through chain alignment in a liquid-crystalline phase, H. Sirringhaus et al., Appl. Phys. Lett. 77, 406 (2000); http://dx.doi.org/10.1063/1.126991.
- Annealing effect of polymer bulk heterojunction solar cells based on polyfluorene and fullerene blend, J-H. Huang et al., Org. Electronics, 10, 27–33 (2009), doi:10.1016/j.orgel.2008.09.007.
- Hole mobility effect in the efficiency of bilayer heterojunction polymer/C60 photovoltaic cells, A. Macedo et al., Appl. Phys. Lett. 98, 253501 (2011); http://dx.doi.org/10.1063/1.3601476.
 F8T2 MSDS Sheet
F8T2 MSDS Sheet