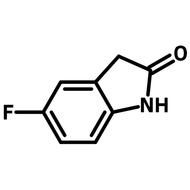
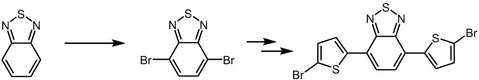
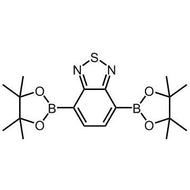
4,7-Dibromo-2,1,3-benzothiadiazole
CAS Number 15155-41-6
Chemistry Building Blocks, Dibromo Monomers, Heterocyclic Building Blocks, Monomers
High purity monomer for organic semiconductor synthesis
Used for applications in LEDs and photovoltaic devices
4,7-Dibromo-2,1,3-benzothiadiazole, CAS number 15155-41-6, is the building block or monomer for organic semiconductors synthesis in the application of light-emitting diodes and photovoltaic devices. Intermediate for the synthesis of i.e. PCDTBT and PCPDTBT.
4,7-Dibromo-2,1,3-benzothiadiazole can be synthesised by using 2,1,3-benzothiadiazole as starting materials to react with bromine in hydrobromic acid. Further bromination of 4,7-Dibromo-2,1,3-benzothiadiazole affords 4,7-bis(5-bromothiophen-2-yl)benzo[c][1,2,5]thiadiazole, a red monomer leading to our pilot polymer PCDTBT which can be used for OPV devices. 4,7-Dibromo-2,1,3-benzothiadiazole is also used for the synthesis of PCPDTBT, a good candidate for the polymer solar cell devices.
General Information
| CAS Number | 15155-41-6 |
| Chemical Formula | C6H2Br2N2S |
| Molecular Weight | 293.97 g/mol |
| Synonyms | Dibromo Benzothiodiazole Dibromo-BT |
| Classification / Family | Benzothiodiazole, PCPDTBT, Semiconductor synthesis, Low band gap polymers, Acceptors, Organic Photovoltaics, Polymer solar cells |
Chemical Structure

Product Details
| Purity | >95% |
| Melting Point | 187 °C - 190 °C |
| Appearance | Light yellow crystals |
Characterisation
MSDS Documentation
 4,7-Dibromo-2,1,3-benzothiadiazole MSDS sheet
4,7-Dibromo-2,1,3-benzothiadiazole MSDS sheet
Literature and Reviews
- Introduction of Perylene Units for Enhanced Interchain Interaction in Conjugated Polymers for Organic Photovoltaic Devices, K. Ji-Hoon et al., Macromolecules, 45(5), 2367-2376 (2012)
- Fluorinated Copolymer PCPDTBT with Enhanced Open-Circuit Voltage and Reduced Recombination for Highly Efficient Polymer Solar Cells, S. Albrecht et al., J. Am. Chem. Soc., 134, 14932-14944 (2012)
Related Products
We stock a wide range of monomers available to purchase online. Please contact us if you cannot find what you are looking for.
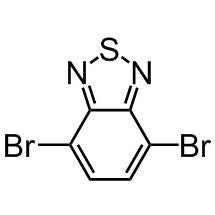
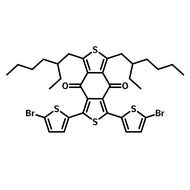
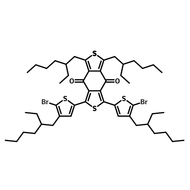
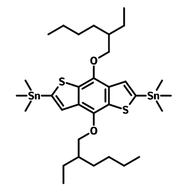
![2-Ethylhexyl 4,6-dibromo-3-fluorothieno[3,4-b]thiophene-2-carboxylate](http://www.ossila.com/cdn/shop/products/ptb7-monomer-b361-ossila-chemical-structure.png?v=1648818400&width=190)
![2,5-bis(trimethylstannyl)-thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/2_5-bis-trimethylstannyl-thieno-3_2-b-thiophene_structure.jpg?v=1504193831&width=190)
![2,6-dibromo-4,4-bis(2-ethylhexyl)-4H-cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/4Hcyclopentadithiophene.jpg?v=1431610575&width=190)
![2,7-Dibromo-9,9-bis[3,3'-(N,N-dimethylamino)-propyl]fluorene](http://www.ossila.com/cdn/shop/products/dibromo-fluorene-diyl-bisdimethylpropan-amine.jpg?v=1431610994&width=190)
![3,6-bis(5-bromothiophen-2-yl)-2,5-bis(2-octyldodecyl)pyrrolo[3,4-c]pyrrole-1,4(2H,5H)-dione](http://www.ossila.com/cdn/shop/products/bisbromothiophenyl-bisoctyldodecylpyrrolo-dione.jpg?v=1431611190&width=190)
![4H-Cyclopenta[1,2-b:5,4-b']dithiophene](http://www.ossila.com/cdn/shop/products/cyclopentadithiophene_12a14774-f96a-4ebc-a0d0-2c5483da9180.jpg?v=1445441165&width=190)
![Benzo[1,2-b:4,5-b']dithiophene-4,8-dione](http://www.ossila.com/cdn/shop/products/benzo-dithiophene-dione.jpg?v=1437904702&width=190)
![Thienothiophene, Thieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/thienothiophene.jpg?v=1431611114&width=190)
![Thieno[3,2-b]thiophene-2-carbonitrile](http://www.ossila.com/cdn/shop/products/thienothiophene-2-carbonitrile.jpg?v=1439548051&width=190)
![DTT, Dithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/dtt-chemical-structure.png?v=1653477307&width=190)
![3,6-Dibromothieno[3,2-b]thiophene (TT36)](http://www.ossila.com/cdn/shop/products/3-6-dibromothienothiophene-chemical-structure.png?v=1653663075&width=190)
![2,3,5,6-Tetrabromothieno[3,2-b]thiophene](http://www.ossila.com/cdn/shop/products/Tetrabromo-thienothiophene-chemical-structure.png?v=1665673773&width=190)
![2,6-Dibromodithieno[3,2-b:2′,3′-d]thiophene](http://www.ossila.com/cdn/shop/products/Dibromodithienothiophene-chemical-structure.png?v=1666702461&width=190)
![2,5-Dihydro-3,6-di-2-thienyl-pyrrolo[3,4-c]pyrrole-1,4-dione](http://www.ossila.com/cdn/shop/products/2_5-Dihydro-3_6-di-2-thienyl-pyrrolo_3_4-c_pyrrole-1_4-dione-chemical-structure-dpp.png?v=1667321819&width=190)
![6,9-bis(5-bromo-4-(2-butyloctyl)thiophen-2-yl)dithieno[3,2-f:2',3'-h]quinoxaline](http://www.ossila.com/cdn/shop/products/bisbromo-butyloctylthiophenyl-dithienoquinoxaline-chemical-structure.png?v=1669202898&width=190)
![Indolo[3,2-b]carbazole](http://www.ossila.com/cdn/shop/products/Indolocarbazole-chemical-structure.png?v=1670495077&width=190)
![10,15-Dihydro-5H-diindolo[3,2-a:3',2'-c]carbazole](http://www.ossila.com/cdn/shop/products/Dihydro-diindolocarbazole-chemical-structure.png?v=1670502109&width=190)
![Indolo[2,3-a]carbazole](http://www.ossila.com/cdn/shop/products/indolo-2-3-a-carbazole-chemical-structure-title.png?v=1678288567&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]propane (BAPP)](http://www.ossila.com/cdn/shop/products/bapp-chemical-structure-title.png?v=1679403349&width=190)
![2,2'-Dimethyl[1,1'-biphenyl]-4,4'-diamine](http://www.ossila.com/cdn/shop/products/2-2-dimethyl1-1-biphenyl-4-4-diamine-chemical-structure-title.png?v=1680597662&width=190)
![2,2-Bis[4-(4-aminophenoxy)phenyl]hexafluoropropane (4-BDAF)](http://www.ossila.com/cdn/shop/products/4-bdaf-chemical-structure-title.png?v=1681225583&width=190)
![1-[2-(Trifluoromethyl)phenyl]imidazole](http://www.ossila.com/cdn/shop/files/1-2-trifluoromethylphenylimidazole-chemical-structure-title.png?v=1682593257&width=190)